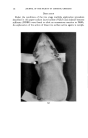
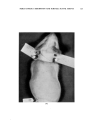
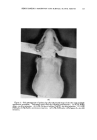
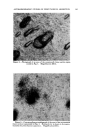
136 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS cated by the fact that at relatively high levels they cause primary irritation when applied alone during the first stage of ten applications. However, when applied again during the second stage of three applications no eczematous response is observed on healed skin. When NiSO4 is given along with NaLS or DDBS during both stages the eczematous dermatitis is observed in the second stage. An im- portant observation is that NaLS alone at the low level of 0.1 per cent shows no primary irritation effect but does spark an eczematous response to NiSO4 late in the first stage and again in the second stage of the multiple application technique. In each case where the nickel response was elicited with NaLS or DDBS, histological analysis showed nickel to be present in the pilosebaceous apparatus, dermis and in some instances the basal layer of the epidermis. This deep penetration of nickel was not observed histo- chemically when NiSO• was applied alone or in the presence of surface active agents failing to elicit any eczematous response. The eczematous reaction observed during the first stage to NiSO• coupled with relatively high levels of NaLS and DDBS is essentially one of primary irritation. However, the irritation response evident during the second stage on skin completely healed from the first stage effects can hardly be called primary irritation in view of the fact that NiSO4 alone and NaLS or DDBS, alone, were without effect. It is believed that NaLS and DDBS in some way increases skin susceptibility to nickel. The guinea pigs used in this study were demonstrated to be moderately sensitive to nickel as de- termined by the Landsteiner-Jacobs intracutaneous injection technique (7). In an effort to explain the eczematous response noted with NiSO4 in com- bination with NaLS or DDBS, the role that acanthosis contributes must be considered. It was observed that both NaLS and DDBS have a decided acanthotic effect on guinea pig skin. The fact that NiSO• was found to penetrate readily through such thickened skin and cause the typical eczematous response raises the possibility that the acanthosis is in some way involved in provoking the nickel reaction. Baer and co-workers (6) sug- gested that acanthosis perhaps makes skin more reactive to allergenic and primary irritant eczematogenic substances and more sensitizable to aller- gens. As mentioned previously repeated vpplications of soap also had the reac- tion of thickening skin but did not provoke the nickel reaction. This may be due to the insoluble nickel soap formed which does not penetrate skin. The possibility also exists that NaLS and DDBS in provoking the nickel reaction have the effect of denaturing epidermal protein, which may result in the easier passage of the metal sensitizer through the epidermis. In this connection the recent work of Harrold (8) would tend to support such a view. Hartold presented data in which he related the effects of different surface active agents in denaturing keratin as reflected in the release of
PERCUTANEOUS ABSORPTION AND SURFACE ACTIVE AGENTS 137 sulfhydryl groups. NaLS was observed to have the strongest denaturing effect on keratin, followed closely by DDBS. Soap and a nonionic agent showed only slight denaturing action. It is well to point out that the concentrations of surface active agents employed in this study were considerably higher than one would expect to encounter in household products under use conditions. For example, in many commercial granulated detergents the surfactant level is about 20 per cent and at the 0.25 per cent normal use concentration of the product, the active level is actually 0.05 per cent. This level is much below the con- centrations studied in this work. Also, the 0.5 per cent level of NiSO4 employed in inducing eczematous eruptions is relatively high when one considers any of the practical over- tones. Upon reducing the level of NiSO4 to 0.1 per cent in the presence of 0.5 per cent DDBS no nickel reaction was observed. The practical significance of the differences noted between surface active agents is difficult to assess in view of the rather stringent testing conditions employed. Certainly one can speculate that individuals hypersensitive to common household and other environmental contaminants probably react to these materials more readily in the presence of certain surface active agents. This thought has been expressed by Suskind (9) and other in- vestigators. CONCLUSION Sodium lauryl sulfate and to a lesser extent, sodium dodecylbenzene- sulfonate applied topically to guinea pigs under the conditions of a two stage multiple application test were observed to provoke the eczematous action of nickel sulfate and also its deep penetration into the skin. Both surface active agents have an acanthotic effect on guinea pig skin and the significance of this action on nickel penetration and irritation is discussed. It is noteworthy that soap at 8 per cent concentration also has an acanthotic effect on guinea pig skin but does not elicit a nickel response. The anion- ics, Igepon A and Igepon T, two nonionics, and one cationic did not elicit the skin response to nickel. ztcknowledgment: The authors wish to acknowledge the technical assist- ance of Mr. Vincent F. Borselli in the maintenance and treatment of the guinea pigs used in this study. REFERENCES (1) Duemling, N. W., •trch. Derrnatol. and Syphilol., 43, 264 (1941). (2) MacKee, G. M., Sulzberger, M. B., Herrmann, F., and Baer, R. L., 5 e. Invest. Dermatol., 6, 43 (1945). (3) Nilzen, A., and Wikstrom, K., •tcta Dermato-IZenereol., 35, 292 (1955). (4) Kvornung, S. A., and Svendsen, I. B., •7. Invest. Dermatol., 26, 421 (1956). (5) Skog, Erik, •tcta Dermato-IZenereol., 38, 1 (1958). (6) Baer, R. L., Rosenthal, S. A., and Sims, C., y. Invest. Dermatol., 27, 249 (1956). (7) Landsteiner, K., and Jacobs, J., 5 e. Exptl. Med., 61, 643 (1935). (8) Hartold, S. P., 5 e. Invest. Derrnatol., 52, 581 (1959). (9) Suskind, R., 7. •trn. Med. •tssoc., 163, 943 (1957).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)