SYSTEMIC TOXICOLOGICAL REACTIONS 119 So the problem, really, is to determine what levels of exposure can be con- sidered safe. TYPES OF E•'•'ECTS Percutaneous toxicity can be important from the standpoint of both acute and chronic effects. Acute toxicity is easier to measure, and, in general, the results are easier to interpret. If the animals die following a single application of the test material, it may reasonably be concluded that a toxic reaction has occurred there is then the usual problem of deciding how likely (or unlikely) it is that any human could ever receive a remotely com- parable exposure. On the other hand, in studying the possible cumulative action of a material, one is usually cotfronted with much more subtle changes in the animal organism in this case the importance of the induced changes must be related to the dosage and frequency of application that induced them then a decision can be made as to the possibility of similar human exposure. Most of this discussion will relate to the possible chronic effects of various compounds, but reference will be made to some materials with which acute toxicity could be the major problem. Although methodology in the study of skin absorption is not a primary concern of this paper, criteria that are applied in the estimation of hazard deserve mention. First, of course, are the gross symptoms of poisoning that may be observed in experimental animals these include any deviations from normal, in behavior or appearance, that are easily recognized, such as anorexia, listlessness, loss of weight, and, as an extreme, death. Gross and microscopic study of tissues may also reveal evidence of pathologic changes, even though no symptoms were seen in the living animals. Useful infor- mation can be obtained by chemical or radiochemical determination of absorption, excretion, and deposition in tissues, especially if the material applied is known to be a cumulative poison. There is one drawback to this approach: if the assay method used is not very sensitive, one may fail to find the compound in the body, even if finite amounts have been ab- sorbed application of a more sensitive analytical procedure may then give some cause for concern about a potential hazard. Obviously, the danger is not increased by an improvement in methodology, but we may present ourselves with a more difficult problem in interpretation. Conse- quently, the demonstration of absorption by means of analysis is likely to be helpful only when there is a considerable background of information on the toxicity of the test material from other standpoints the heavy metals, especially lead and mercury, may be cited as examples in this category. In these cases it may be possible to conclude that the amounts absorbed are too
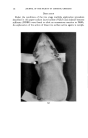
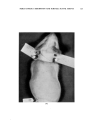
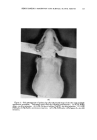
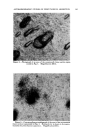
120 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS small to produce cumulative effects under normal conditions, or, conversely, that the compound is deposited in tissues in quantities that may represent a real hazard. Otherwise, with materials of unknown toxicity or known to be of low toxicity, we do not learn much from a demonstration that a given number of micrograms can be found in the blood, urine, or liver after a gram has been rubbed into the skin. TYPES OF MATERIAL STUDIED A look at the literature revealed that there is information, of one kind or another, on the absorption through the skin of a considerable number of materials more or less related to cosmetics. For many of these, there is evidence that absorption can occur but that toxicity does not result from dosage levels that might be encountered in normal usage. In addition, it can reasonably be concluded that the common ingredients of cosmetic prod- ucts (including emollients, surface active agents, etc.) are safe from the standpoint of percutaneous toxicity even though results of specific tests may not have been reported. In many cases these compounds are known to be nontoxic by other routes of administration, so that there should be no hazard even if absorption occurs. This position is often supported by many years of usage with no indication of a safety problem. A few materials that may be of particular interest have been selected for special consideration these have been chosen either because of some particular properties that seem of importance, or because the investigations themselves are worthy of special comment. BORIC AOtD An example of the borderline area between cosmetics and drugs is to be found in boric acid, which has been widely used in a variety of products for many years. It is said that boric acid has been used in mouthwashes at levels of 2 to 4 per cent, in eye lotions at 1.5 per cent, and in henna shampoos at $ per cent. A patent covers a deodorant stick containing 25 per cent boric acid. The USP ointment, containing 10 per cent boric acid, is used in dermatologic practice, and a saturated aqueous solution (about 4.5 per cent) has been used in wet dressings. The most common application is at a level of $ per cent in talc as a dusting powder, especially for the diaper region in babies. The buffering, rather than' the antibacterial, action seems the more important in the latter case. There is no doubt that boric acid may be rather toxic under certain cir- cumstances. Fatal poisoning has occurred as a result of its absorption into the body by several different routes. Many of the deaths have resulted from accidental ingestion when boric acid solution was mistakenly substi- tuted for water or glucose solution in the preparation of infants' formulas, but some can be attributed to absorption of the compound through the skin.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)