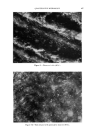
486 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 1A 1E lB IF IC 1G IH Figure 1.--Stabilized (1A, lB, 1C, 1D) and unstabilized (1E, IF, 1G, 1H) lather (bright field 125X). 1A and 1E represent fresh foam lB and 1F show foam after 4 min. 1C and 1G show foam after 15 min. 1D and 1H show foam after 25 min.
QUANTITATIVE MICROSCOPY 487 products of the industry, such as creams, sprays, lotions, polishes, powders and emulsions. The purpose of the microscopic examination is not only to study the effect of one upon the other. It also is applicable to basic re- search and to research directed toward product improvement and toward ways of achieving the same purpose in a more economical way. Microscopic techniques are by their very nature optical techniques, and the optical properties of the above mentioned specimens are used to de- scribe their state and appearance or the dynamics of any change or inter- action. A number of optical properties lend themselves to quantitative determination. First, there are the directly measurable material char- acteristics: spectral absorption and spectral reflectivity, refraction and dispersion, birefringence, dichroism, and all the related polarization-optical material constants. Finally, there are the spectral emission characteristics of fluorescing and phosphorescing materials. Determinations of one or more of these characteristics can be used to obtain quantitative information on derived parameters, which directly describe product properties or the dynamics of certain processes. A typical example is the measurement of optical path differences in an interference microscope, which can be used to measure skin smoothness directly, as will be described later on. Sometimes it may become necessary to create and define new parameters in order to obtain a measurable quantity which shows a direct correlation to the process under examination. This technique will be illustrated with the aid of a practical example. It is undesirable for a lather used for shaving purposes to collapse rapidly. It is well known that collapsing can be prevented by adding a stabilizing agent. The photomicrographs (Fig. 1) show two samples of shaving lather, one with and the other without stabilizer. The lather was prepared in a thin layer, and the pictures were taken over a period of ap- proximately 30 minutes. Hardly any change occurs in the stabilized lather. The unstabilized preparation collapses completely. During this process a great many changes can be observed. The wall strength of the bubbles decreases, the bubble diameter increases, and the shape of the bubbles changes. Also, the number of bubbles per unit area decreases remarkably. Any one of these parameters may, of course, be used to describe the col- lapsing behavior of the lather. However, these various parameters may have different differential sensitivities to the effect under investigation, in this case to a change in the amount of stabilizing agent added. In other words, one should try to find that parameter which shows the highest coefficient of correlation to the variable under study. In the specific example here it was the relative number of bubbles per unit area which showed better than the other parameters the high rate of collapse of the unstabilized lather, as shown in Fig. 2. Once such a pa- rameter has been found, one can easily derive other quantities, such as a
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)