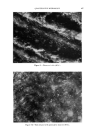
488 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS rate of collapse coefficient or time constants and one can study the effects of temperature, water hardness, and similar external influences upon these derived parameters. By plotting one of these values against the amount of added stabilizer, one obtains numerical values for the minimum amount of stabilizer which will satisfy the requirements for product quality, e.g., by guaranteeing foam stability over a period of time not longer than is 'E • 50 0 I•) 20 t min. Figure fl.--Collapsing behavior of two samples of lather. absolutely necessary. The stabilizer may be an expensive additive, and such studies can contribute to keep its use at a cost-saving minimum. The previous example has purposely been exaggerated to demonstrate the usefulness of newly created derived quantities. In other cases, th determination of some of the optical properties leads directly to numerical values. Below, the sensitivities of a number of methods will be discussed. The numerical values given here are intended to indicate the possibilities and the limitations of the procedures. They represent a rough estimate based on reasonable values from experimental experience. In many cases, higher accuracies and higher sensitivities could be reached with more dab-
QUANTITATIVE MICROSCOPY 489 orate techniques employed by those who in the patent literature are so aptly described as "skilled in the arts." MICROSPECTROPHOTOMETRY Spectrophotometry plays an important role in almost every aspect of chemical research. When the specimen has microscopic dimensions, the sample compartment of a standard spectrophotometer may be replaced by a microscope and the sample cell by the microscope slide. Such in- struments are called microspectrophotometers. They have found wide- spread application in histochemistry, where minute amounts of substances are traced by their absorption, or where microchemical tests can be carried out even within individual cell components by means of color reactions. Microspectrophotometers permit identification of substances by means of their spectral absorption characteristics. Determinations of concentra- tions of the relative and sometimes even of the absolute amount of chemical compounds are possible. Sensitive microspectrophotometers have meas- uring areas as small as 1 X 1 /•. There are also microspectrophotometers for measurements in the ultraviolet spectral range (1,2). An estimate of the sensitivity of these methods leads to the following values. It can be assumed that a minimum absorption of 5% is sufficiently high above the noise level to exclude statistical errors. It shall further- more be assumed that the specimen has a geometric thickness of 10u and that the molecular coefficient of extinction at the maximum of the extinction curve has a value of 15,000 cm.2/mMole. This is an average value. Dyes very often have molecular coefficients of extinction as high as 35,000 cm.•'/mMole, whereas substances of a simpler structure such as trypto- phane have coefficients of around 8000 cm.=/mMole. The concentration can be calculated from the formula, ] = [:•lO-•cd in which I is the transmitted intensity, I0 the incident intensity, d stands for the geometric thickness of the specimen expressed in centimeters, c is the concentration expressed in Mole/10acm. a, and e is the molecular coeffi- cient of extinction, which has the dimension cm.=mMole. Rearranging leads to 1 ,r0 •d log f = o The minimum detectable concentration, using the values given above, would then be 1 100 Mole 15,000 X 10 X l0 -4 log • = 1.2 X 10 -a X 10 a cm Assuming a measuring area of 4 X 4u and a section thickness of 10/•, the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)