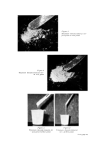
IR SPECTROSCOPY TO TOILET ARTICLES AND HOUSEHOLD PRODUCTS 611 was less than that in combar A. The strong absorptions at 1185 and 1060 crn -• indicated the presence of an ionic sulphonate, which was not attached to an aromatic ring since there were no absorptions at 1610 and 840 cm-L The absorptions at 1730 cm -• showed the presence of an ester grouping and comparison with spectrum 7a showed that the detergent in cornbar B was the sodium isethionate ester of a fatty acid. The ATR spectra of three toothpastes are shown in Fig. 2. The absorptions at 1120, 1052, 1002 and 940 crn -• for A and B were due to 1800 1400 I000 650, WAVELENGTH CPI -t Figure oe. ATR spectra of toothpastes. glycerol while that at 1640 cm -• was due to water. The very broad strong absorption at 1410 crn -• and the doublet at 860 and 885 cm -• in the ATR spectrum of toothpaste B (Fig. 2b) showed that it contained a large amount of a carbonate which was not present in the other toothpaste. The spec- trum of C showed a low concentration of carbonate together with sorbitol shown by the broad weak absorptions at 980 to 1120 crn-L Fig. $ records the ATR spectra of three liquid washing-up products. The absorptions at 1090, 1050 and 890 crn -• in the ATR spectrum of A (Fig. $a) showed that it contained ethanol, while the absorptions at 1180,
612 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 1130 and 1020 cm -t indicated that a dodecylbenzene sulphonate was present. The absorptions at 1200 cm -•, due to the asymmetrical SO•. stretching vibration, and 930 cm -x showed that an ionic sulphate was also present. The other characteristic absorption shown by such sulphates, which occurs at ca. 1050 cm -x, would be hidden by the ethanol absorption at 1050 cm -•. It was concluded therefore that the active ingredient in A was a mixture of a sulphonate and a sulphate. The shoulder at 1250 cm -x showed that an aromatic ether grouping was present, which may be associated with the ionic sulphate. The alternative would be that this grouping was associated with an ethoxylated alkylpheno1 present as a nonionic. I•OO 14oo IOOO 650 WAVELENGTH CH -1 Figure 3. ATR spectra of washing-up liquids. Since there was no absorption at 890 crn -• in the ATR spectrum of B (Fig. 3b) it did not contain ethanol. The absorptions at 1180, 1130, 1040, 1015 and 840 cm -• showed that this product contained a dodecyl- benzene sulphonate. As the intensity of the absorption at 1015 crn -• was less than that of the absorption at 1040 cm -•, by comparison with reference sample, there were no toluene or xylene sulphonates present. The absorp- tion at 1110 crn% due to aliphatic ether groupings, and those at 1630 and 1550 cm% due to an amide group, suggested that this product contained an ethoxylated fatty amide as a nonionic.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)