PERCUTANEOUS ABSORPTION OF TRICLOSAN 215 REFERENCES (1) Black, J. G., Sprott, W. E., Howes, D. and Rutherford, T. Percutaneous absorption of hexachlorophene. Toxicology 2 127 (1974) (2) Curley, A., Hawk, R. E., Kimbrough, R. D., Mathenson, G. and Finberg, L. Derreal absorption of hexachlorophene in infants. Lancet Aug 7 296 (1971). (3) Ulsamer, A. G., Yoder, P. D. and Marzulli, F. N. Determination of hexachlorophene in human and experimental animal tissue. Toxicol. Appl. Pharmacol. 22 276 (1972). (4) Lyman, F. L. and Furia, T. Toxicology of 2,4,4Mrichloro-2•-hydroxy diphenyl ether. Ind. Meal. 38 45 (1969). (5) Black, J. G., Howes, D. and Rutherford, T. Percutaneous absorption •tnd metabolism of Irgasan DP300. Toxicology 3 33 (1975). (6) Vickers, C. F. H. Dam, reservoir or filter. Trans. St. John's Hosp. Dermatol. Soc. 59 10 (1973). (7) Feldman, R. G. and Maibach, H. I. Regional variations in percutaneous absorption of •4C-cortisol in man. J. Invest. Dermatol. 48 181 (1967). (8) Maibach, H. I., Feldman, R. J., Milby, T. H. and Serat, W. F. Regional variation in per- cutaneous penetration in man. Pesticides. Arch. Environ. Health 23 208 (1971). (9) Bartek, M. G., Labudde, J. G. and Maibach, H. I. Skin permeability in vivo: comparison in rat, rabbit, pig and man. J. Invest. Dermatoi. 58 114 (1972). (10) Tregear, R. T. Physical Femctions of Sicin p. 12 (1966) (Academic Press, London).
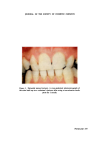
J. Soc. Cosmet. Chem. 26, 217-225 (1975) i•) 1975 Society of Cosmetic Chemists of Great Britain A parametric test to measure cleaning power of toothpaste the W. B. DAVIS and D. A. REES* Presented on 26-30th •lugust 1974 in London at the IFSCC I•IIIth International Congress on 'Cosmetics--Quality and Safety' organized by the Society of Cosmetic Chemists of Great Britain. Synopsis---A sensitive IN VIVO method was developed to quantify the cleaning power of dentifrices. Volunteers built up natural STAIN on their TEETH over a period of 5 weeks by replacing their usual TOOTHPASTE with a non-abrasive paste. The estimated •o area of the incisors covered by stained pellicle was observed to increase, especially towards the end of this build-up period. The natural stain was progressively removed by a series of 10 s BRUSHING periods using an electric TOOTHBRUSH and the pastes to be tested. The logarithm of the area of stain was plotted against the duration of brushing with the test paste and found to conform to a linear trace. Thus an equation for stain removal may be of the form S=ae t't with the coefficient b being an expression of the ability of the test paste to remove stain. INTRODUCTION Over thirty years ago, Manly (1) observed that the 'brown pellicle' which formed regularly on the teeth of some people who did not use denti- frice abrasives could be readily removed by one or two brushings with a dentifrice grade calcium carbonate or phosphate but not by brushing with a dry or moistened brush without a dentifrice. Lobene (2), and later Van Abb6 et al. (3) who slightly modified Lobene's technique, employed direct visual observation to assess the removal of stained material from teeth using a toothpaste under controlled brushing conditions. Wilkinson and Pugh (4) used both photographic recording and * Beecham Products Applied Research and Evaluation Laboratories, RandalIs Road, Leather- head, Surrey. 217
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)