192 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Brand G (6 samples): 6 with 25TDA 1 with 2AF 5 with 3AF 4 with 4AF 1 with 2NPFD 1 with AN 2 with 24DAA Brand H (12 samples): 6 with 25TDA 6 with 2NPFD 6 with RES 1 with HCH 2 with AN 1 with CRES Brand I (9 samples): 7 with 25TDA 7 with 3AF 1 with 2NPFD 7 with RES Brand J (2 samples): 2 with 25TDA 2 with 3AF 2 with HCH 1 with AN Unidentified spots: 8 Conclusions: No p.phenylenediamine was found. 2.5.Toluylenediamine and Resorcinol were the most important compounds. The 3 isomeric aminophenols are frequently used. 2.nitro.p.phenylenediamine is much used, 4.nitro.o.phenylene- diamine less. Of minor importance are: e-naphthol, hydroquinone, chloro- resorcinol, 2.4.diaminoanisol. Abbreviations: 25TDA = 2.5.Toluylenediamine 2AF = 2.Aminophenol 3AF = 3.Aminophenol 4AF = 4.Aminophenol 2NPFD = 2.Nitro.p.Phenylenediamine 4NOFD = 4.Nitro.o.Phenylenediamine RES = Resorcinol CRES = 2.Chlororesorcinol HCH = Hydroquinone AN = e-Naphthol 24DAA = 2.4.Diaminoanisole. THE ANALYSIS OF SUNTAN PREPARATIONS Suntan preparations may contain several classes of potentially hazardous sub- stances to man, such as sunscreens (uv absorbers), browning agents (mainly dihydroxyacetone), and local anaesthesics (for instance ethyl p.aminobenzoate or benzocaine and lidocaine). These cosmetics are applied on large surfaces of the
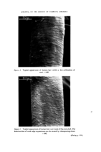
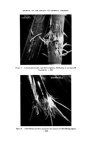
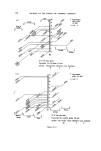
RISK-BEARING SUBSTANCES IN COSMETICS 193 body, by old and by young people, and intensively during several weeks annually. At that time the skin is exposed to the sun, the air and the water. It is possible that under such rigorous conditions of use the toxicological hazards of the active components will increase. To get an idea of the diversity in the use of the sunscreens in particular, analytical data of some 66 suntan preparations (24 brands) was collected. The identification was made mainly by tlc-glc matching with industrial sunscreens as references, and combined with chemical group reactions of two important classes of sunscreens. By the kind cooperation of the chemical industry, some 27 sun- screens were collected for reference analysis (Table VI). This number, however, is far below the possibilities mentioned in literature (22, 23) (Table VII). The list considered is given in Tables VI and VII. Table VI. List of reference industrial sunscreens Firmenich Solprotex 1, 2, 3 Givaudan Parsol mcx Parsol ultra Givtan F Laserson/Sabety Ecranosol Solecran Felton Sunarome GAF Uvinul ms40 Uvinul n539 Uvinul 400 Norda/Schimmel Angstrol Filtrosol A, B, Triple Naarden Solisoline A, B Merck Eusolex 3573 Eusolex 4360 Eusolex 161 Eusolex 232 Rhone-Poulenc Rhoditan L Dragoco Prosolal S9 Merck Eusolex 6653 Salicylates. 2.ethylhexyl p.methoxycinnamate. Mixture, not known. 2.ethoxyethyl p.methoxycinnamate. Salicylates. Unknown. 2.ethylhexylsalicylate. 2.hydroxy.4.methoxy.5.sulphonic acid. benzo- phenone. 2.ethylhexy1.2X.cyano.3.3 X.diphenylacrylate. 2,4 x.dihydroxybenzophenone. Salicylates. Salicylates. Unknowil. 4.phenyl.2.carbonicacid isoctyl ester.benzo- phenone. Unknown. 3.4.dimethoxyphenyl-glyoxylate Na. 2.phenylbenzimidazoline. Salicylates. Mixture of phenylacrylic and oxybenzoic esters. Dibenzalazin. Thin-layer chromatography of sunscreens In the tlc system used, several dark spots from a single extract usually appeared under uv light. To detect a sunscreen spot the following procedure was used. The spot is scraped off, extracted with 3 ml of methanol, filtered, and the uv
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)