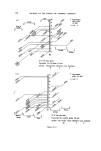
194 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table VII. Inventory of sunscreens mentioned in the literature (22, 23) A. Para amino benzoic acid and esters: ethyl, butyl, propyl, glyceryl. B. N.N.Dimethyl para amino benzoic acid ester: isoamyl. C. Ortho aminobenzoic esters (anthranilic acid esters): methyl, menthyl, phenyl, benzyl, linalyl, cyclohexenyl, bornyl, isobornyl. D. Salicylic acid esters: menthyl, homomenthyl, phenyl, benzyl, glyceryl, dipropylene glycol, 2.ethylhexyl. E. Cinnamic acid esters and derivatives: 2.ethylhexyl p.methoxy cinnamate, benzyl cinnamate, 2.ethoxy ethyl p.methoxy cinnamate, menthyl cinnamate, benzyl p.methoxycinnamate, i.butyl salicyl cinnamate, propyl p.methoxycinnamate, butyl cinnamoyl pyruvate, ct-phenyl cinnamonitrile, 2.ethylhexyl 2X.cyano 3.3X.diphenylacrylate. F. Dihydroxycinnamic derivatives: umbelliferone, methylumbelliferone, methyl aceto-umbel- liferone. G. Trihydroxycinnamic acid derivatives: esculetin, I].methyl esculetin, dafnetin, glycosides of esculin and dafnin. H. Benzophenone derivatives: 2.4X.dihydroxybenzophenone, 4.phenylbenzophenone, carbonic acid isooctylester. 1. Quinine oleate, tannate, stearate, bisulphate. J. Coumarin derivatives: 7.hydroxy-, 7.methyl-, 3.phenyl. K. Digalloyltrioleate. L. Dibenzalazine. M. Dibenzalacetophenone, benzalacetone. N. Benzimidazolen, phenylbenzimidazolon sulphonic acid Na. O. 2.Phenyl benzoxazole, methylnaphthoxazole. P. 2.Phenyl benzothiazole. Q. Stilbene. R. 2.Acetyl bromo indazole. S. 8.Hydroxy quinoline, 2.phenyl quinoline. T. Butyl carbityl 6Lpropyl piperonyl ether. absorption spectrum determined between 250 and 320 nm. At such a low con- centration level (approx. 30 gg per 3 ml = 10 ppm) a sunscreen still has an absorption maximum at near 300 nm. Other dark spots of the chromatogram, such as from preservatives, will not have an appreciable absorption at 300 nm (see Fig. 9). Experimental Extraction: Dissolve 1 g of the sample in 4 ml methanol. Heat gently. Add 1 g Na2SO4 exsicc. Mix thoroughly. Use upper methanol layer for tlc or glc. Reference solution: Use 1 • w/v solutions of reference sunscreens in methanol. If the sunscreen is not soluble in methanol, dissolve by adding small amounts of water to the mixture until a clear solution is obtained. Tic-plates: Glass plates covered with a 0.25 mm layer of silica GF (with fluorescent indicator), and activated at 105øC for « h.
RISK-BEARING SUBSTANCES IN COSMETICS 195 o o o o o o 8 X X X X X • Sunscreenspots (Absorption meXo et .300 nm) E=I.O .... E_--/'O _ _ -. __ •_o.5 2 _ 280 :300 280 :300 280 300 280 300 280 300 nm nm nm nm nm Figure 9. Detection of a sunscreen spot on a tic plate. Scrape off the spot from the glass plate. Extract with 3 ml of methanol. Filter. Determine the uv spectrum of the solution in a 1 ml quartz cell, between 250 and 320 nm. If at this low concentration level (approx. 10 ppm) an appreciable absorption occurs at 300 nm, the tlc spot is a sunscreen. Examples: tlc of the samples no. 37, 35, 21, 24, 25 uv spectra of the spots. Solvent system' Diisopropylether-n. Hexane-Acetic acid (20 + 80-t- 1 vol). Unsaturated tank. Spots' For the extracts or reference solutions 5-10 pl. Visualization' Uv 254 nm. Gas-liquid chromatography of sunscreens Gaschromatographic conditions. Column: 6}/o Apiezon L and 10•o KOH on Chromosorb W 60-80. 150 cm diam. 0.25 inch, copper. Temperature column: isotherm 230øC. Injection port: 240øC. Detector: 250øC (flame ionization). Carrier gas' Nitrogen. Extraction: as under tic. Reference solution: as under tic. Injection: •-1 [tl.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)