204 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS mixtures. The calculated results are tables in Table XII in which it can be con- cluded that for these levels of concentration cyclohexane is acceptable as a general internal standard in our procedure. Method of determination of linearity of glc response. The determination of the correction factors F, by simple injection of standard mixtures of which the com- position is known exactly was first obtained. Several standard mixtures were made before and stored in 100 ml glass aerosol bottles without dip tubes, so that they can be used over and over again. The analysis of such a standard mixture and the calculation of the correction factors F is given in Fig. 12. The correction factors should be determined daily for optimal reliability. They might vary a little, depending on the slightly different glc conditions day by day. Since every injection on the gas chromatograph takes 30 min for total response, sampling in the intermediate transfer vessels can be done in the meantime, in- cluding the addition of the internal standard. Of each sample 20 Ixl amounts should be injected and the •o calculated from the integrated data as shown in Fig. 13. By this single injection a total quantitative analysis of the propellants solvents is accomplished within half an hour. A simple analytical control has been devised for each determination, namely by summation of the found •o, including the • of the non-volatile residue which should be determined separately by simple gravimetric analysis. If the sum of the percentages differs more than 5•o from the theoretical 100•o, the analysis should be repeated, including the sampling procedure. This simple control has been proved valuable during our analytical survey, as fault analysis can be avoided. Results The results of the analysis of the 60 hairspray aerosols are pictured in Fig. 14, which shows histograms of the levels found in the samples. ACKNOWLEDGMENTS The author wishes to thank A.M. de Roos, Director of the Food Inspection Service of Enschede, for his promotion of this work and for the permission to publish. The author gratefully acknowledges the technical assistance of G. J. Runder- voort, T. D. Wesseldijk-Osinga and J. Rooselaar. (Received: 27th March 1974)
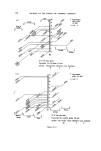
2O 3O 4O 5O 6O 7O 8O 9O RISK-BEARING SUBSTANCES IN COSMETICS Recorded Integrated retention peak areas times in seconds 119 235055 3 II 82022 486 478216 881 10675 1097 142462 1204 61066 1508 14721 From the recorded retention times the following compounds are identified: PI2 PII Aceton Dichl. meth. CYCL. HEX. Ethonol i Propan ol The correction factors for that day were,. PI2 = I. 53 PII = 1.55 Act: 0-82 DCM: I' I0 EtOH: 0-78 i PrOH = O- 87 205 • ecorded retentio times in seconds , 4.8E 881 1097 1204 1508 Figure 13. Example of the glc analysis of a hairspray aerosol sample no. 34. The weight per cent were calculated with the formula % = (Ax. Fx/As). (s/m). 100 % in which Ax = integrated peak area of compound 1 Fx --= correction factor of compound 1 A s = integrated peak area of Cyclohexane s = weight in grams of Cyclohexane m = weight in grams of the sampled mixture. The following data were obtained: Propellant 12 .... 35.5 % Propellant 11 .... 12.7 % Acetone .... 39.3 % Dichloromethane .. 1.2 % Ethanol .... 4.9 % isoPropanol .... 1.3 % For the purpose of analytical control, we determined the non- volatile residue by gravimetric analysis and found--Residue .. 1.9 % Total found ................ 96.8 % This sum of per cent differs less than 5% from the 100•o, so the analysis is acceptable.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)