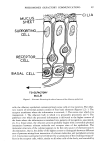
62 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table 1. Time required for the reaction to come to completion at each temperature. Temperature Time Raw materials (øC) (hr) TiO2 (54.0 m2/g) 600 4 800 1 1000 1 TiO= (25.0 m•/g) 600 20 800 4 1000 1 TiO2 (8.5 m•/g) 600 20 800 20 lOOO 20 also shows that the time for the reaction to come to completion becomes longer with decreasing Ss= of the TiO= used. This is understandable based on the consideration that the reaction rate becomes faster as the particle size of the starting material decreases, since the decrease in particle size causes the lowering of the melting point or of the lat- tice defects of crystals. This was noted by Kelvin and Herring (5,6). The reaction time did not change with the mixing ratio of Ti and TiO2. As well known, the reaction can be made faster by raising the calcination temperature, but the latter causes the aggregation of particles by sintering. Since higher temperature causes stronger aggregation, the reaction should be conducted at the lowest tempera- ture possible within the conditions of the time stipulated. Thus, it is reasonable that each of the TiO= of P-25, P-110, or #328 grades can be calcined at 600øC for 4 hr, 800øC for 4 hr or 1000øC for 20 hr, respectively. TESTS FOR PIGMENTS For application as a black pigment in cosmetics, the degree of blackness and tinting strength of the pigment should first be examined. These conditions were studied so as 24 •, 23 -- 22 o TiO2 (A)/ 2 3 4 n Figure 3. Change of L-value as a function of n for the synthesized TinO•n-•
LOWER TITANIUM OXIDE 63 70 •Ti 2504-f- Ti02 '"", (•..,,. Iron Oxide Black O-- Iron Oxide Black © •0 -- 0"•---•" Ti2504 • • • • • 1•2 2 4 6 8 10 m2/g Figure 4. Change of L-value as a function of the SN2 of synthesized Ti2.,504 for the l0 per cent mixture of Ti2.504 and TiO2 to obtain the most useful lower titanium oxide. These results are shown in Fig. 3-4. These data suggest that the blackest pigment is obtained at n = 2.5 regardless of the type of TiO2 used and when n is fixed the pigment having the largest SN2 shows the highest in tinting strength. Thus, a pigment having a higher tinting strength can be synthesized using a TiO2 having the smaller particle size. Use of TiO• P-25 as a starting material is advantageous because of the ease of synthesis. Crystallographically, Ti•.•O4 is a mixture of Ti•O.• and Ti.•Os. VISCOSITY OF A SLURRY IN WHICH THE PIGMENT WAS DISPERSED Figure 5 shows the plots of viscosity of the slurry in which the obtained pigment was dispersed versus the ratio of the pigment/TiOz. In the figure, FeaO4 is also plotted for the convenience of comparison. Clearly, the influence of the new black pigments on the viscosity was smaller than that of FeaO4. This fact is important when formulating different shades of cosmetics in a given product line. Should the viscosity change with 130 --Fe30• T•• x 110 •. 90 • x x ß • 70 • a .-'/ Ti2 •04 (B) • ....... o 50 ........ •o Ti2504 (C) 30• __ • , Ti02 25 50 75 Ti2504 (white) weight ratio (black) Figure 5. Change of viscosity as a function of mixing ratio of Ti•.•O• and TiO• for the slurry
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)