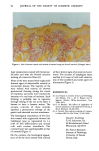
ANALYSIS OF PAINT-ON ARTIFICIAL NAILS 71 Table V Quantitative Interpretation of NMR Spectrum of Typical Artificial Nail Liquid Parts by Weight Relative Molar Substance Intensity Found Taken Relative Error Isobutyl methacrylate Ethyl methacrylate Ethylene glycol dimethacrylate N,N-dimethylamino-p-toluidine 42.9 46.6 45.0 3.6% 49.4 43.1 45.0 4.2% 4.7 7.1 10.0 29% 3.1 3.2 3.2 0 Hx o 1 2 Et nB iB Fr Td 8 9 1• Eg 1 12 13 Minutes Figure 10. Gas Chromatogram of Methacrylate Ester Mixture.
72 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Where a minor component must be determined by taking the small difference between two much larger numbers, as for ethylene dimethacrylate in this example, the accuracy suffers. Where a minor component can be determined separately from its own distinct and relatively sharp spectral peaks, as can the amine in this example, the accuracy is generally quite good. Replicate measurements on five different resin mixtures gave an estimated 95% confidence interval for a single determination of the weight percentage of a given constituent of 3% absolute (determined value +/--3) when the value must be obtained by solving a set of simultaneous linear equations. When the peaks for a constituent were isolated, so its relative molar intensity could be independently determined, the estimated 95% confidence interval for a single determination was 0.5-3% relative, depending on the nature and complexity of the mixture. The impurities in commercial methacrylate esters, and the extensive overlap in the spectra involved, make difficult the attainment of greater precision without more sophisticated techniques. The use of such techniques is scarcely warranted in the present case, especially in view of the availability of chromatographic methods. The gas chromatographic conditions recommended by Black (3), and a similar set used by him in an analysis of Lee Nails liquid (10), were found to be inconvenient when monomers less volatile than butyl methacrylate were present. Even ethyl methacrylate gives badly tailing peaks, and the retention time of, for example, tetrahydrofurfuryl methacrylate is of the order of 113 minutes. The conditions used in the present work resulted in better peak shape and more convenient retention times, even for monomers as high boiling as triethyleneglycol dimethacrylate. Figure 10 is the chromatogram of a mixture of six of the methacrylates, and one amine, found among nail extender formulations. Peak separations and shapes are obviously adequate for good quantita- tive determinations. Table VI summarizes the interpretation of this chromatogram, using ethyl methacrylate as convenient standard for the determination of relative sensitivity factors. Replicate measurements on test mixtures gave an estimated 95% confidence interval for a single determination of the weight percentage of a given Table VI Gas Chromatogram of Some Components of Artificial Nail Liquids Rel. Wt. Rel. Area Rel. Peak Substance in Mixt. of Peak Sensitivity A Air Hx Hexanes Me Methyl Methacrylate 0.999 1.004 1.005 Et Ethyl Methacrylate (1) (1) (1) iB Isobutyl Methacrylate 0.996 0.970 0.974 nB n-Butyl Methacrylate 1.002 0.985 0.983 Td N,N-Dimethyl- p-Toluidine 0.181 0.167 0.923 Fr Tetrahydrofurfuryl Methacrylate 1.070 0.924 0.864 Eg Ethylene glycol Dimethacrylate 0.994 0.739 0.743
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)