342 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 2O 15- 10- _ 20 15- 10- _ i IIi I 0.1 PC liposomes 1.0 10.0 PClChol liposomes lOO - 80 - 60 - 40 - 20 - o I i i i i I 100.0 ii I i -- i i i i i i i 0.1 1.0 10.0 100.0 100 8O 6O 4O 2O Size (I.rm) Figure 1. Size distribution of fresh phospholipid liposome preparations prepared by RPE protocol 2• Top: PC liposomes bottom: PC/cholesterol 9:1 molar ratio. phologies were observed by cryo-TEM, including unilamellar and multilamellar lipo- somes, vesicles encapsulating smaller vesicles and tubular structures (Figure 2). SELECTION OF A SEPARATION METHOD The centrifuged-column separation method, dialysis, and the discontinuous Ficoll ©
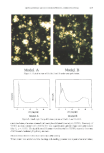
ENCAPSULATION INTO LIPOSOMES 343 .: , O:,lla O.1}am Figure 2. Cryo-TEM micrographs revealing several different vesicular morphologies observed in PC lipo- somes prepared by RPE protocol 2. Long, elongated structures are shear-induced artifacts of the observation method. The large honeycomb structure is part of the mounting apparatus. Arrows indicate inverted cubic phase. Magnification: 66,000x. density gradient separation method were tested for their ability to separate free inulin from liposomes. For each method, two test preparations were evaluated. A 2% w:v solution of inulin containing 14C-Carb-I was used to follow free inulin, and 14C-DPPC- labeled PC liposomes were used to evaluate liposome recovery. For the centrifuged-column method, the radiolabeled inulin solution was tested with Sephadex G-50 fine and Sephadex G-25 fine. At the first pass, 86% of total radiolabel was retained by the Sephadex G-50 column, whereas only 39% was retained by the Sephadex G-25. After two passes, 96% of radiolabel was retained by the Sephadex G-50 column and 65% was retained by the Sephadex G-25. Sephadex G-50 was therefore selected for the rest of the study and tested for its ability not to retain radioactivity associated with 14C-DPPC liposomes. Contrary to the results of other investigators (13,33,34), radiolabel recovery from the column eluant was extremely poor (less than 9%). Since most investigators reporting good results have worked with smaller lipo- somes (100-400 nm in diameter), we tested the hypothesis that the poor recovery was due to the large diameter of the liposomes studied. This was done qualitatively by sonicating liposome preparations, a common physical treatment to decrease liposome size. There was a positive correlation between sonication time and radiolabel recovery, indicating that the centrifuged-column method was not appropriate for large liposomes. Such limitations have been reported elsewhere (35). However, sonication or other physi- cal treatment aiming at decreasing liposome size was not a preferred alternative in our study since we felt it could yield leakage and significantly decrease EE. For this reason, the centrifuged-column method was not pursued further.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)