402 JOURNAL OF COSMETIC SCIENCE straightforward to formulate, these emulsions can be sensitive to extremes in formulation pH and salt content. The rheology and break characteristics of these emulsions can be far different than other emulsions - for better or worse. More often, water-soluble polymers are used to augment a primarily steric stabilized emulsion. Liquid crystalline phases are two dimensionally organized multilayers of lipophilic emulsifier. Most often used to build viscosity, stability, and pleasing rheology in steric stabilized systems, these phases can be used as the primary emulsion stabilization mechanism with excellent effects. If used as the primary emulsifier, these can stand the help of a small amount of polymer to assist stability. Even with the help, these emulsions are not very tolerant to excesses of salts or low pH. With a better understanding of the emulsion options available including their strengths and limitations, the cosmetic chemist can better serve their target market (and thus rise to fame and fortune.)
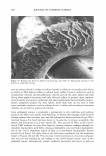
2004 ANNUAL SCIENTIFIC SEMINAR LIQUID CRYSTALS: How TO USE THEM. WHY THEY ARE OUR FRIENDS. Robert Lochhead, Ph.D. The School of Polymers and High Pe,formance Materials University of Southern Mississippi, Hattiesburg, MS 39406-0076 Surfactant aggregation in aqueous solution leads to well-defined nanostructured assemblies that exhibit spherical, rod-like or sheet-like structures 4 and since the discovery of liposomes by Bangham in the 1960's S . these lyotropic mesomorphic phases have held promise in applications that require precise nanostructured materials. Liquids have no long-range order and perfect solid crystals have three-dimensional order. Liquid crystals are materials that have intermediate order that is one-dimensional or two-dimensional order. There are two principal classes of liquid crystals: thermotropic and lyotropic. Themotropic liquid crystals are the types commonly found in displays: (watches, Effect of Molecular Structure on Micelle Shape and Size Packln& factor• v/al, Packing fador, vi� . 1/3 . -..,.,,._ ... ___ , .... ..,,_ Packln1 ractor, IJJ,•fal.: l/l The molecular shape cnn be approximated by a cyllnder --+ --+ calculators) and lyotropic liquid crystals are found in cosmetic, pharmaceutical and household formulations. Lyotropic liquid crystals are formed by self-assembly of amphipathic surfactant molecules. The concept of packing into supramolecular assemblies having precise structure can be understood by consideration oflsraelichvili's 'packing factor' definitions. 1 The packing factor is defined as the volume at the hydrophobic tail group divided by the volume of the cylinder subtended from Packing factor, ,•/al e , 1/3 the head group along the entire length of the --•--• .. --- molecule . Pocklna lllctor, 1/J ,·tal c 1/l .-...:: ... , ................. .. As the packing factor increases the curvature of the self-assembled micelles decrease and this causes a change in micelle shape from spheres to rods to layers to inverse rods and eventually to inverse spheres. 403
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)