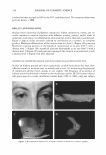
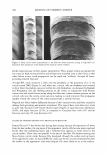
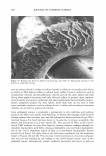
ASSESSMENT OF OCULAR IRRITANCY 325 Table III Correlation Coefficient (r value) of Fluorescein Staining Between Categories Liquid Baby Eye makeup Powder eye Facial makeup Shampoo wash remover Mascara shadow cleanser Liquid makeup 1.00 Shampoo 0.92 1.00 Baby wash 0.91 0.95 1.00 Eye makeup remover 0.94 0.88 0.82 1.00 Mascara 0.85 0.88 0.77 0.97 1.00 Powder eye shadow 0.88 0.92 0.99 0.77 0.71 1.00 Facial cleanser 0.92 0.95 0.98 0.79 0.71 0.98 1.00 wash, and powder eye shadow elicited the most similar fluorescein staining patterns at post-instillation intervals. Human ocular instillation is an effective and safe in vivo methodology for the assessment of cosmetic irritancy. Ocular irritation elicited by the evaluated test materials varied markedly by product category, with respect to the type (subjective reports, inflamma tion, and fluorescein staining), duration, and ocular tissue involvement, demonstrating that these factors are important considerations for the prediction of the ocular irritancy of a test material. ACKNOWLEDGMENT The authors acknowledge Julie R. Meyers for her valuable assistance in writing this paper. REFERENCES (1) N. E. McCain, R. R. Binetti, S. D. Gettings, and B. C. Jones, Assessment of ocular irritation ranges of market-leading cosmetic and personal-care products using an in vitro tissue, Toxicologist, 66 (1-S), 243 (2002). (2) P. A. Jones, E. Budynsky, K. J. Cooper, D. Decker, H. A. Griffiths, and J. H. Fentem, Comparative evaluation of five in vitro tests for assessing the eye irritation potential of hair-care products, Altern. Lab. Anirn., 29, 669-692 (2001). (3) Y. Gao and B. E. Kanengiser, Observation of ocular irritation in human subject, IOVS, 41, 5257 (2000). (4) J. D. Burdick, Y. Gao, B. E. Kanengiser, J. C. Merrill, and J. W. Harbell, Comparative assessment of two eye area cosmetic formulations through evaluation of alternative eye irritation methods relative to endpoints measured in a human clinical sub-acute study design, Society of Toxicology 2003 Annual Meeting. (5) J. H. Draize, G. Woodward, and H. 0. Calvery, Methods for the study of irritation and toxicity of substances applied topically to the skin and mucous membranes,]. Pharrnacol. Exper. Therapeutics, 78, 458-463 (1944). (6) J. S. Friedenwald, W. D. Hughes, and H. Hermann, Acid-base tolerance of the cornea, Arch. Ophthal rnol., 31, 279-283 (1994). (7) U.S. EPA, Primary eye irritation study in pesticide assessment guidelines, subdivision F hazard evaluation: Human and domestic animals, EPA Guidelines 1982, 51, 81-84 (1982). (8) U.S. EPA, Toxic substance control act test guidelines, part 798: Health effect testing guidelines. Subpart B-General toxicity testing, Fed. Reg. 1985, 50, 39398.
J. Cosmet. Sci.J 55, 327-341 Quly/August 2004) Evaluation for collagen products for cosmetic application YONG PENG, VERONICA GLATTAUER, JEROME A. WERKMEISTER, and JOHN A. M. RAMSHA W CSIR0 Molecular Science} 343 Royal Parade, Parkville, Victoria 3052, Australia. Accepted for publication January 15, 2004. Synopsis Collagen is an important component for cosmetic formulation, where it is an effective natural humectant with high substantivity. Commercial collagen preparations have a wide range of properties. In the present study, various techniques have been used to examine three distinct commercial collagens that illustrate the range of properties that are available. The usefulness of the various techniques for assessing collagen quality and batch-to-batch variation is discussed. The results indicate that there are several simple, cheap, and effective methods, such as gel electrophoresis, that provide excellent information on collagen quality. The appropriate selection of tests allows informed decisions on the choice of which collagen preparation to use to provide the desired functionality and shelf life of a formulation. INTRODUCTION Collagen has become a valuable and well used component in cosmetic formulation, where it provides significant benefits. In particular, it is an effective natural humectant (1,2) as a result of the extensive, ordered hydration network that surrounds the molecule (3), in combination with its high substantivity to the skin surface. A wide variety of collagen preparations is now available that are suitable for cosmetic formulations. These preparations vary considerably in their composition and properties. Quality testing to allow informed decisions on the choice of which collagen preparation to use is therefore important so as to ensure that the preparation provides the desired functionality. Testing should also be capable of monitoring potential batch-to-batch variations that could also affect product performance. Also, it should indicate whether the collagen is in its native, triple-helical form or is present in its denatured form, gelatine. Native collagen is characterized by a triple-helical structure, in which three extended left-handed polyproline II-like helical chains are supercoiled into a right-handed triple helix, linked through interchain hydrogen bonds (4,5). The triple-helical conformation requires a distinctive amino acid sequence, in particular glycine as every third residue, Address all correspondence to John A. M. Ramshaw. 327
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)