JOURNAL OF COSMETIC SCIENCE 426 compounds that are related to the main psychoactive ingredient in marijuana: Δ9- tetrahydrocannabinol (3). Phytocannabinoids are present in a large variety of plants and can interact with cannabinoid receptors or share chemical structure with cannabinoids, or both (4). The endocannabinoid system comprises endogenous ligands such as the anan- damide, enzymes, transporter apparatus, and two known receptors (CB1 and CB2). The various downstream effects of the cannabinoids are mediated through these two G protein– coupled receptors. The selective activation of cannabinoid receptor 2 is devoid of psycho- active side effects associated with cannabinoid receptor 1 stimulation. Skin infl ammatory reaction is characterized by redness, swelling, heat, and pain. Even if this is an important host defense mechanism against invading pathogens, persistent or over-infl ammation can result in skin damage. Distribution of CB1 and CB2 in human skin is described in many cell types and structures, and many studies have reported the promising role of canna- binoids in the treatment of dermatologic conditions, among them skin infl ammation (5–10). A downstream signaling mechanism of cannabinoid receptor 2 activation is the β-endorphin release, contributing to the peripheral analgesic effect (11). β-endorphin is a proopiomelanocortin (POMC)-derived opioid neuropeptide, and on a molar basis, β-endorphin is 18–33 times more potent than morphine (12). CB2 receptor activation alone is enough for attenuation of the neuropathic pain, by attenuating infl ammation, and changes in the interleukin signal (13). A large body of evidence now exists to sub- stantiate that certain endocannabinoids activate transient receptor potential vanilloid 1 (TRPV1). TRPV1 is a key receptor of various sensory phenomena (pain, heat, and itch). It is found in nonneuronal cell types, including human skin epidermal keratinocytes (14–17). TRPV1 also acts as a nociceptive sensor and can potentiate the infl ammatory process. Indeed, elevated TRPV1 expression was identifi ed in UV-irradiated photo-aged and intrinsically aged skin (18,19). Cannabinoids also exert potent anti-infl ammatory effect however, the mechanism by which cannabinoid receptor type 2 reduces infl amma- tion and promotes tissue repair in the course of human skin wound healing is still not completely clear. Several plants produce molecules with cannabinoid activity (20), such as β-caryophyllene (BCP), a phytocannabinoid with a strong affi nity to cannabinoid re- ceptor 2 (21), but not to cannabinoid receptor type 1 (22). BCP is one of the major active components of essential oils derived from a large number of spice and food plants. Patchouli oil has a variety of pharmacological activities, including anti-infl ammatory (23,24). BCP as a plant volatile compound is commonly found in patchouli leaves, with other sesqui- terpenes (25,26). Activity of BCP can be partially described as a cannabinoid receptor 2 agonist, suggesting its role as an anti-infl ammatory agent (27). In this study, we investigated the key functions of cannabinoid signaling to control local infl ammation responses in the skin, with CB2 receptor agonist AM1241, antagonist AM630, and a patchouli extract. Our fi ndings suggest that our patchouli extract possesses a soothing effect via the modulation of the CB2 receptor pathway. MATERIAL S AND METHODS PLANT MA TERIALS AND EXTRACTION The aeri al parts comprising the stem and leaves were collected in the fi eld from sustain- able farming in Colombia, dried, and ground by cryogenic grinding. Pure plant extract was obtained using a supercritical CO2 extraction technique, including ethanol as a cosolvent.
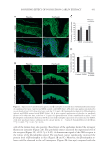
SOOTHING EFFECT OF POGOSTEMON CABLIN EXTRACT 427 This process allowed us to obtain a nonpolar fraction (containing essential oil), and a polar fraction containing molecules such as phytosterols, fl avonoids, and fatty esters. Pure extract was diluted in octyldodecanol, and was used at 1% on ex vivo skin and at 0.001% on cultured keratinocytes. ANTIBODI ES Primary antibodies used were anti-CNR2 (Thermo Scientifi c, Waltham, MA), anti–β- endorphin (LSBio, Seattle, WA), anti–interleukin receptor 1 (IL1R1) (Rockland, Limerick, PA), anti–interleukin 6 signal transducer (IL6ST) (Santa Cruz Biotechnology, Heidelberg, Germany), and anti-TRPV1 (Thermo Scientifi c). Alexa Fluor® coupled secondary anti- bodies were used (Molecular Probes, Eugene, OR). REAGENTS Sel ective an d competitive antagonists of cannabinoid receptor 2: AM630 and selective agonist of cannabinoid receptor 2: AM1241 were purchased from Sigma Chemical Co. (St. Louis, MO). AM630 and AM1241 were applied on skin biopsies at 1 mM, 3 h (topical and culture medium application) before UVB irradiation with 200 mJ/cm2 (Bio-Link Irradiator 254 nm, Fisher Scientifi c, Illkirch, France). Lipopolysaccharide (LPS) (Sigma- Aldrich, St. Louis, MO) was used at 0.5 mg/mL overnight. CELL CULTURE Normal human epithelial keratinocytes were isolated from skin obtained from plastic surgery of healthy females who had given written informed consent. Keratinocytes were cultured in keratinocyte serum–free medium, with provided human recombinant epider- mal growth factor and bovine pituitary extract (Gibco, Auckland, New Zealand), and 0.1 mg/mL Primocin™ (Invivogen, San Diego, CA). β-ENDORPHIN S YNTHESIS IN CULTURED KERATINOCYTES Human keratino cytes were grown in tissue culture dishes (100 mm). After the addition of the patchouli extract, plates were incubated for 24 h with the patchouli extract at 0.001% [diluted in 0.001% dimethyl sulfoxide (DMSO)]. Cells were harvested, and β-endorphin was measured by enzyme-linked immunosorbent assay (Elabscience, Houston, TX). HEMOTOXYLIN AND EOSIN STAINING Slides containi ng 4 μm skin paraffi n sections were deparaffi nized and rehydrated in the following baths: 2 × 2 min in xylene, 2 × 2 min in 100% ethanol, 1 × 2 min in 95% ethanol, 1 × 2 min in 80% ethanol, and 1 × 5 min in H2O. Hematoxylin staining: 1 × 3 min hematoxylin and rinsed in water for 1 × 5 min. Eosin staining and dehydration:
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)