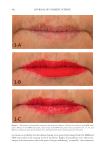
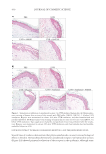
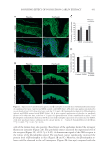
JOURNAL OF COSMETIC SCIENCE 434 epidermis and some cells of the dermis. Application of the patchouli extract signifi - cantly increased the protein level of β-endorphin and the release by keratinocytes. Our results suggest that β-endorphin release contributes to the soothing effects of the patchouli extract. Our results are consistent with those of a previous study, where AM1241 stimulated β-endorphin release from immortalized human keratino- cyte HaCaT cell line (33). It is also possible that other mediators, in addition to β-endorphin, might be released after the activation of CB2 receptors, contributing to the soothing effect. TRPV1 acts as a key peripheral integrator of pain, itch, and heat sensation in skin. TRPV1 was found in human skin and might be associated with infl ammation. Pharmacological blockade of TRPV1 has recently emerged as a potential novel therapeutic possibility in managing infl ammatory diseases (34). In our experimental model, using UVB- and LPS-induced infl ammation, skin treated with the patchouli extract showed less- ened production of TRPV1 and pro-infl ammatory cytokine receptors, including IL6ST and IL1R1. These results demonstrate that the activation of the CB2 receptor, at least in part, might participate in keratinocyte protection through the inhibition of cytokine receptors. The patchouli extract helped prevent the activation of TRPV1 and interleukin infl ammasome pathway, and, in parallel, limited the formation of sun burn cells, as demonstrated by the suppression of tissue damage (Figure 1A and B). Likewise, the patchouli extract formulated at 1% helped to improve skin sensitivity by reducing stinging sensation induced by the activation of TRPV1 via capsaicin (Figure 4). The use of the patchouli extract associated with protective action although CB2 receptor activation could be a promising strategy for the treatment of sensitive skin. CONCLUSIONS In summary, our results confi rm that CB2 agonist has strong anti-infl ammatory activity in the human skin exposed to UVB or LPS. This effect, at least in part, is associated with the reduction in TRPV1, IL1R1, and IL6ST levels and the release of the β-endorphin opioid. A Pogostemon cablin extract (patchouli) containing the phytocannabinoid BCP, and other anti-infl ammatory molecules present in the patchouli extract such as the polyphenols, was associated with a reduction in the level of infl ammatory markers, via cannabinoid Figure 4. Evaluation of skin sensitivity by stinging test with capsaicin after 28 of cream applications. Decrease in capsaicin-induced stinging sensation after face application of a cream containing patchouli extract at 1% (n = 10 mean ± SEM p d 0.05).
SOOTHING EFFECT OF POGOSTEMON CABLIN EXTRACT 435 receptor 2 modulation. This extract could be used to lessen skin discomfort, especially in case of fragile skin and itching sensation. A CKNOWLEDGMENTS We are grateful to Laura Mouret for the analysis of bioactive compounds present in the pa- tchouli extract, and to Ludivine Mur and Alexia Lebleu for the technical assistance. We wish to thank Anne Clay Viardot for reading the manuscript and for the English language review. Patents: Ashland owns patent applications related to a method for obtaining an extract of patchouli leaves and cosmetics uses thereof, published under numbers FR3091993A1 and W02020156981. Funding: This research was funded by Ashland Inc. Author contributions: Florian Labarrade, Yolene Ferreira, Gilles Oberto, and Catherine Gondran conceived and designed the experiments, and the methodology Florian Labar- rade, Armelle Perrin, Yolene Ferreira, and Gilles Oberto performed the experiments and analyzed the data and Florian Labarrade wrote the paper, with Catherine Gondran revising it critically. Corine Morel was in charge of patchouli extract procurement and analysis. Catherine Gondran and Karine Cucumel supervised the study and handled project administration. All authors read and approved the fi nal manuscript. REFERENCES (1) A. Oláh, A. G. Szöllősi, and T. Bíró, The channel physiology of the skin, Rev. Physiol. Biochem. Pharma- col., 163, 65–131 (2012). (2) T . C. Theoharides, J. M. Stewart, A. Taracanova, P. Conti, and C. C. Zouboulis, Neuroendocrinology of the skin, Rev. Endocr. Metab. Disord., 17, 287–294 (2016). (3) P . Pacher, S. Bátkai, and G. Kunos, The endocannabinoid system as an emerging target of pharmaco- therapy, Pharmacol. Rev., 58, 389–462 (2006). (4) J . Gertsch, R. G. Pertwee, and V. Di Marzo, Phytocannabinoids beyond the Cannabis plant – do they exist? Br. J. Pharmacol., 160, 523–529 (2010). (5) S . Ständer, M. Schmelz, D. Metze, T. Luger, and R. Rukwied, Distribution of cannabinoid receptor 1 (CB1) and 2 (CB2) on sensory nerve fi bers and adnexal structures in human skin, J. Dermatol. Sci., 38, 177–188 (2005). (6) J. Zhang, L. Chen, T. Su, F. Cao, X. Meng, L. Pei, J. Shi, H. L. Pan, and M. Li, Electroacupuncture in- creases CB2 receptor expression on keratinocytes and infi ltrating infl ammatory cells in infl amed skin tissues of rats. J. Pain, 11, 1250–1258 (2010). (7) J. L. Zheng, T. S. Yu, X. N. Li, Y. Y. Fan, W. X. Ma, Y. Du, R. Zhao, and D. W. Guan, Cannabinoid receptor type 2 is time-dependently expressed during skin wound healing in mice, Int. J. Leg. Med., 126, 807–814 (2012). (8) C. D. Río, E. Millán, V. García, G. Appendino, J. DeMesa, and E. Muñoz, The endocannabinoid system of the skin. A potential approach for the treatment of skin disorders, Biochem. Pharmacol., 157, 122–133 (2018). (9) J. S. M ounessa, J. A. Siegel, C. A. Dunnick, and R. P. Dellavalle, The role of cannabinoids in dermatol- ogy, J. Am. Acad. Dermatol., 77, 188–190 (2017). (10) R. Mi l ando and A. Friedman, Cannabinoids: potential role in infl ammatory and neoplastic skin diseases, Am. J. Clin. Dermatol., 20, 167–180 (2019). (11) F. Ga o . L. H. Zhang, T. F. Su, L. Li, R. Zhou, M. Peng, C. H. Wu, X. C. Yuan, N. Sun, X. F. Meng, B. Tian, H. L. Pan, and M. Li, Signaling mechanism of cannabinoid receptor-2 activation-induced β-endorphin release, Mol. Neurobiol., 53, 3616–3625 (2016).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)