164 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Wittgenstein has found that by increasing the pH, dihydroxyacetone may react with most of the amino acids. Actually, how much of the dihydroxy- acetone applied to the skin is complexed by the amino acids and how much is absorbed as dihydroxyacetone? Sweat from colored areas still complexes dihydroxyacetone to produce color. How can we increase the range of reactivity of the locally available dihydroxyacetone in relationship to the amino acids available in the superficial skin ? As yet we do not know the answer. According to our studies, only a small amount of the available amino acid in keratin is used in the complex. Even with maximum con- centrations of 90 per cent dihydroxyacetone and repeated applications, we seem to reach a saturation point in the development of color which we cannot change either quantitatively or qualitatively. Other investigators have found the same result (3, 4). From our clinical studies on over 280 patients ($), it is apparent also that this complexing does not interfere with any obvious skin function, such as, reaction to infection and, cur- iously, even to yeast infection, or to other forms of reactivity in the skin. We are now studying the vaginal flora under local dihydroxyacetone and also the changes in morphology of the fungi in contact with dihydroxyace- tone in their media including media containing only amino acids. There- fore, since we do have available dihydroxyacetone reactive groups, carbonyl and available amino acids, we should be able to get more amino acid com- plexing. We are continuing to study this problem. We have found recently that we can inhibit color formation on skin by using sodium thiosulfate with dihydroxyacetone or calcium chloride in dihydroxyacetone. You are all familiar with the formaldehyde inhibition of the color complexing (1, 4). Phenol does not inhibit the development of color, neither does glycerol. In our preliminary experiments, dihydroxyacetone phosphate did not produce a color. Moreover, we obtained no color with dihydroxyacetone and N,l-carbobenzoxy arginine. In our experience, glyceraldehyde showed weak coloring (from production of dihydroxyacetone?). Glyoxal shows more rapid development and more homogenous coloring. The material, however, is highly irritating and sensitizing and should not be used in cosmetics. pH of the dihydroxyacetone reaction is of interest. Bandelin (6) has indicated that no color is produced by solutions of pH 8.3 or over. How- ever, in freshly prepared borate buffer solutions of pH 9.5, we have pro- duced color. As the dihydroxyacetone reacts with buffer (and pH is lowered) no coloring develops. It is true, of course, that the pH of the solution applied is not the pH at the reaction site. The coloring on the skin changes the pH of the colored area very little. For example, in one patient, pH changed from 6.4 to 6.7. The other phase of the study which is of more concern to you is the
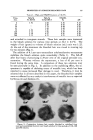
TOXICOLOGIC AND CLINICAL INVESTIGATIVE STUDIES 165 study of the color complex itself. Here, we feel that you have much more data than we. We have felt from the beginning that although the use of callus was superior tO gelatin, yet this is of value only for early pilot testing studies. It is in the living normal skin where the dynamic study of the coloring lies. For testing series, both individuals who are known to color rapidly and those who color slowly are used. Coloring should be observed in skin not only urlder controlled conditions, but also with the skin micro- scope under moderate magnification (7) and with light filtered through the Wood's Filter, 3620 A., for there are characteristics of fluorescence at vary- ing stages in the development of the color (1). It is interesting to watch the development of the color in forms of little particles scattered, not in uniform fashion, in superficial portion of the skin. The masses are larger, more darkly colored in the more heavily keratinized areas. Microscop- ically, the color masses may be observed well in deparafFinized unstained skin. We have studied this also under polarized light (1). We can watch the loss of color under the skin microscope by the use of bleaching agents, such as sodium hypochlorite, and by the use of sodium bisulfite. Form- aldehyde applied to the skin prior to the application of the dihydroxyace- tone will inhibit color it will not change it after the color has developed. Peeling with Scotch tape, as you know, will remove color and the color can be studied in the tape strips when these are suspended in glycerol and observed under the microscope (1). Color particles were found scattered irregularly through the skin often in the ridges. These color masses ob- served at 80X appeared globular or spherical in outline. The pigment masses were studied also under polarizing microscope and with fluorescent microscopy. No particles were found in the skin appendages. Histo- chemical studies are under way with the relationship of dihydroxyacetone to the amino acids in the skin. The effect of local injections of solutions of dihydroxyacetone in the skin has been studied. One injection, intradermally, of a commercial preparation of dihydroxyacetone into the skin of a volunteer did not cause color to develop in the superficial skin. Injections have been made with 20-90 per cent aqueous solutions of dihydroxyacetone subcutaneously in guinea pigs. No color was observed grossly in the deeper tissue but coloring of the skin and hair above the injection site was observed (? con- tamination). Coloring was studied also with various materials such as scales, hairs, epidermal cysts immersed in solutions of dihydroxyacetone. In addition to studies with microscopy, an analysis of the color was made in human skin by color reflectance by the G.E. Reflecting Electrospectro- photometer. The color was compared to sun tanned areas in the same individual and in other subjects. Report of this has been made elsewhere (8). Color reflectance curves were compared also with solutions of di- hydroxyacetone and amino acids.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)