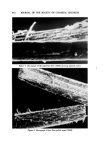
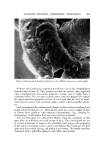
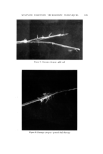
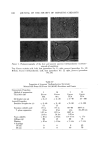
MICROBIAL CONTROL 657 Since deionization resins break down at temperatures above 60øC, hot wa- ter sanitization cannot be employed to reduce the microbial lead within the DI beds. An alternative procedure, employing 2% formalin disinfection, is given in Table I. Table I Formalin Disinfection of DI Resin Beds 1. Close valve between carbon and DI beds. 2. Open all outlet valves in system. 3. Pump 2% formalin solution (1 gal 37% formalin q- 17.5 gal water) into inlet of DI column. Continue pumping until effluent from all outlets gives positive (pink) Schiff test. a 4. Close all valves and allow formalin to remain in the system for 12-18 hours. 5. Flush entire system with tap water until effluent from all taps gives negative Schiff test. 6. Regenerate DI beds. •Test: To 7 ml of effluent add 1 ml of Schiff reagent, mix. Pink indicates a positive reac- tion, colorless indicates negative. Schiff reagent: Fuchsin 0.5 g, sodium bisulfite 9.0 g, conc. HC1 10.0 ml, H, q.s. 500 ml. Prepare fresh daily. Formalin treatment should be utilized when routine microbiological sur- veys of the effluent show a high total count or the presence of a specific or- ganism such as Pseudomonas. If .a carbon bed is present in the water system, it should be isolated by valving prior to the introduction of the formalin. If the piping downstream of the DI column is stainless steel, it may be steamed rather than formalin treated. When the piping downstream of the DI column is PVC, formalin treatment, rather than steam, should be used since PVC will not withstand temperatures in excess of 60øC. Ultraviolet irradiation is commonly used to reduce the microbial level of the water used in product compounding. Ultraviolet radiation (2,400-2,800 A) is relatively easy to use, but even with an optimum system, the effective depth of penetration is never more than 100/xm (7). Dissolved and suspended min- eral and organic content of the water drastically reduce the effect of penetra- tion. Deposits slowly build up on uv lamps and, even with regular cleaning, result in a decay in efficiency between cleanings. Flow rate, contact time, and hours of lamp operation are also critical and exact requirements which need to be established empirically and monitored regularly. For a combination of these reasons, uv radiation tends to be inconsistent and unreliable and is gen- erally not used as a sole means of achieving sterilization. Sterilization of water by heat requires a temperature of 121øC and a pres- sure of 15 psi. The large pressure vessels necessary are not commonplace in production units for cosmetic products. Sometimes lower temperatures (65-
658 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 80øC) are used to reduce the population of gram-negative organisms. Theo- retically, a temperature of 80øC held for 30-45 minutes will kill all vegetative organisms, but not spores. In practice, not even all the vegetative organisms are killed, for there are always pockets of cooler water where microorganisms survive. Condensation droplets falling back into the tank are another source of recontamination. Filtration is often used to achieve sterile or low-count make-up water. Sev- eral filter materials are available that have been termed "bacterial retentive." However, only a filter that removes 100% of all bacteria can be considered a true sterilizing filter, for even if a few viable organisms penetrate, they can soon reinfect an entire filtered batch. Thus, success in this critical procedure hinges on selection of the ideal filter properties. Filter media may be grouped into two broad categories, depth or screen. Screen filters have uniform pore openings and retain particles on their sur- faces, primarily by a sieving mechanism. Examples of screen filters are woven wire mesh or photoetched screens. The most useful characteristic of a screen filter is that pore openings can be controlled during production with such pre- cision that it is possible to specify precisely the maximum size of particles which will pass through the filter. A screen filter is an absolute filter it will retain 100% of all particles larger than its maximum pore size. Depth filters consist usually of fibers, particles, or other fragmented materi- als that have been pressed or bonded together to form a tortuous maze of flow passages. Examples are asbestos pads, ceramic candles, porous sintered glass, and complex laminates of asbestos impregnated with a resinous bonding agent to increase porous density. Although the flow channels in a depth filter can be made coarse or fine, de- pending on the degree of compaction or the size of the fibers, pore size dis- tribution is inherently random and therefore unpredictable. The principal mechanisms of retention are random adsorption and entrapment. Therefore, a filter that is 99% efficient at retaining 0.5-/xm organisms may also retain many that are much smaller, while at the same time passing a substantial number of particles and microorganisms larger than 1 it/All. For very small particles and most microorganisms, adsorption is the most important retention mechanism, since some flow channels invariably are large enough to let them pass. But, because the molecular forces of adhesion are usually strong enough to offset the small viscous drag at the laminar layer of the fluid boundary, a microorganism will adhere if it comes in contact with the filter medium. Unfortunately, some filter manufacturers attempt to assign "absolute" rather than "nominal" ratings to depth filters. This is contradictory because these fil- ters cannot, by their very nature, be absolute even though they may appear so under certain carefully limited conditions (8). But if flow conditions are al-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)