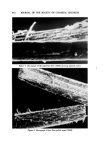
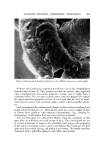
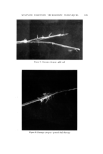
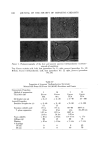
COSMETIC PRESERVATION 669 over prolonged periods will prove the stability and efficacy of the preservative system. Certain components of a for•nulation can enhance the activity of a preserva- tive system. Some common cosmetic ingredients, such as many perfume oils, have antimicrobial properties. Various alcohols, aldehydes, and acids also have antimicrobial activity. Some preservatives are made more effective by the presence in the cosmetic of other ingredients. These enhancing ingredients may have little or no antimicrobial action of their own, but can facilitate antimicrobial activity. Alternatively, enhanced antimicrobial effectiveness may result from the use of two or more preservatives. Synergism in preservation means increased activity resulting from the use of two or more preservatives. In order to constitute synergism, the effect of the combination must be more than additive. Thus, combinations of parabens are superior to the individual parabens. This is generally acknowledged as synergism, although some people maintain that this is strictly a solubility ef- fect, i.e., the parabens are so poorly soluble in water that combinations merely permit a higher total paraben concentration. Another illustration of syner- gism is the combined preservative effectiveness of parabens with Germall©* 115, to be discussed later. Combining parabens with Germall 115 significantly increases the capacity of the preservative system to withstand microbial in- sult, and also widens the range of microorganisms against which the cosmetic is protected. COSMETIC PRESERVATIVES Selection of a suitable preservative or preservative system for a cosmetic must take into account the composition and intended use of the cosmetic. Firstly, the preservative must be compatible with the color and fragrance of the cosmetic, and with all the other components. Above all, there should be no inactivation of the preservative by components of the cosmetic. Secondly, the antimicrobial activity in the specific formulation must be determined. This is done by microbial challenge testing of the cosmetic. Thirdly, antimicrobials are by their nature active against living tissue and must be checked, in the specific formulation, for toxicity, irritation, or sensitization. There is no gain in replacing a microbial hazard with another, possibly worse, problem of toxicity. Fourthly, the preservative must be reasonably stable to storage. A preserva- tive which evaporates, like ethyl alcohol, or breaks down in alkaline medium, or reacts with the components of the cosmetic, as does formaldehyde, might not afford protection a few months later. Lastly, the preservative should not only be soluble in water but also have limited or no tendency to migrate into a nonaqueous phase, where it affords little or no protection. Bruch has listed "some of the more important chemicals which have found general acceptance as preservatives in cosmetic preparations" (9,). For more *Registered trademark of Sutton Laboratories, Inc., Roselie, N.J.
670 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS complete data on specific preservatives, the series by Gueklhorn (3) should be consulted. Ethyl Alcohol Bruch (2) indicates that this compound is active against gram-negative bacteria at concentrations greater than 10%. Most other workers report that at ]east 15% ethyl alcohol is needed in acid solution, and at least 18% in neutral or alkaline media. Actually, even concentrations of 5 or 10% alcohol are use- ful, but these low concentrations are usually only bacteriostatic. Propylene glycol acts in a way similar to ethyl alcohol. Quaternary Ammonium Compounds. These compounds look very good in screening experiments with microbio- logical media although they are relatively ineffective against some gram- negative organisms, including pseudomonads. They have their best activity at about pH 7 to 8, and like hypoehlorites are useful for sanitizing equipment. Unfortunately, quaternary ammonium compounds are irritating to skin and eyes even at concentrations of a few tenths of 1%. They are inaetivated by many ingredients: anionies, nonionies, and proteins. Sorbic Acid Sorbic acid has been used in foods for over 25 years because of its low tox- icity. It discolors somewhat on storage and is only slightly soluble in water (about 0.16% at room temperature). Potassium sorbate, because of its high water solubility, is often used to incorporate sorbic acid. Potassium sorbate it- self, however, causes intense eye irritation. Problems that arise with the use of sorbic acid include: some skin irritation at use dilutions (0.15-0.20%), a pH dependence (half of sorbic acid is ionized at pH 4.8), and poor activity against bacteria. Sorbic acid is often ineffective in the presence of nonionics. It tends to migrate into fats and oils, although not to the extent benzoic acid does. Benzoic acid has a low solubility in water and is therefore usually in- corporated as its sodium salt. Because benzoic acid is a stronger acid, it is limited to use at pH's even lower than those usable with sorbic acid. Benzoic acid has higher activity against bacteria, as well as good activity against yeast and molds, and is less irritating to the skin than sorbic acid. Dehydroacetic acid, as mentioned previously, can be used at somewhat higher pH's than ei- ther sorbic acid or benzoic acid. Hexachlorophene Until recently, hexachlorophene was the most widely used antimicrobial in cosmetics. Its key property is substantivity, the ability to remain on the skin
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)