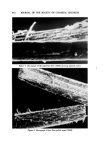
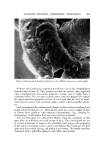
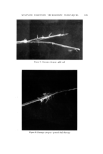
COSMETIC PlqESERVATION 605 contamination. A massive microbial contamination can overwhehn any prac- ticable preservative system. The next three factors are more complex. The pH of a cosmetic oeormulation ought to bc the first consideration in designing a preservative system. Many preservatives become less effective or are unstable at certain pH's. Quater- nary ammonium compounds are more ef[ective at a pH above 7. Mercurials form an insoluble precipitate above pH 8.5. Some common preservatives have an acidic hydrogen. Sorbic acid and benzoie acid, for example, are earboxylic acids. A rise in pH is by definition a decrease in the hydrogen ion concentra- tion. It is obvious oerom the following ionization equilibrium RCOOH • RCOO- + H + that decreasing the hydrogen ion concentration shifts the equilibrium to the right. With increasing pH more of the carboxylic acid preservative is changed to carboxylate anion. Unfortunately, the dissociated, anionic form is not active antimicrobially, so a rise in pH converts the active carboxylic acid to the in- active carboxylate artion. A possible reason for the inactivity of the anion is that microorganism cell walls tend to have a slight net negative charge, and this could repel the similarly charged anion molecules. Phenols also have weakly acidic hydrogens, but since the phenol-phenolate ion equilibrium, ArOH • ArO- + H + tends to be shifted to the left, it takes a higher pH to convert half the phenol to phenolate anion than it does to convert half the molecules of a carboxylic acid to carboxylate anion. Even nonphenolic preservatives may have acidic hydrogens. If they do, then a rise in pH will again remove active, undissociated preservative from the left side of the following equilibrium, Preservative-H • preservative- + H + and generate inactive dissociated anion. An example of a preservative with an acidic hydrogen, which is not a carboxylic acid and not a phenol, is dehydro- acetic acid (DHA). The hydrogen on carbon 3 of DHA (I) is acidic because it is surrounded by three carbonyl groups. OH COCH 3 CH3" •0 / '0 COOR Dehydroacetic acid p-Hydroxybenzoate ester ( D•IA ) ( paraben ) II
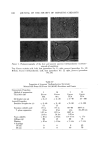
666 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS The general formula for parabens (II) is also shown. The different para- ben members differ only in the alkyl group of the ester: R is methyl in methyl paraben, propyl in propyl paraben, and so forth. Since the earboxylie acid ester is neutral and not acidic, parabens do not have the same pH dependence as benzoie acid. However, parabens are also phenolie, and phenols are •veak acids. Table I Per Cent Undissociated Preservative at Different pH's pH Preservative 4 5 6 7 Sorbic acid 86 37 6 0.6 Benzoic acid 60 13 1.5 0.15 Dehydroacetic acid 95 65 16 2 Methyl paraben 77 63 Table I indicates the percentage of active preservative molecules at several pH's. Even at pH 4, sorbic acid is only 86% in its undissociated, active form. The other 14% is carboxylate anion, which is inactive. It does not matter whether sorbic acid or potassium sorbate is added to a cosmetic. If the cos- metic is at pH 4, the equilibrium of 86% undissociated sorbic acid and 14% ionized sorbate will be established. At pH 6, only 6% of any sorbic acid added is active antimicrobially, and at pH 7, less than 1% is undissociated. At higher pH's the amount of undissociated sorbic acid is negligible. Benzoic acid is a slightly stronger acid than sorbic acid, and the percentages in Table I bear this out. At pH 5, benzoic acid is only of marginal value for preservation since only 13% is in the active undissociated form. Dehydroacetic acid (I) is a weaker .acid than sorbic or benzoic acid, but it is stronger than most phenols. At pH 6, dehydroacetic acid has more active form present than benzoic .acid does at pH 5. At pH 7, where carboxylic acid preservatives contribute almost no antimicrobial action, dehydroacetic acid retains some limited activity. Since the dehydroacetate anion itself is weakly antimicrobial, some cosmetics containing DHA exhibit slight antimicrobial activity even at pH 7. Even though the parabens are not as dramatically affected by pH as are stronger acids, the same principles apply. At pH 7 only two-thirds of the methyl paraben has effective antimicrobial activity at pH 8.5 only about half of the methyl paraben is undissociated and active. The other parabens behave in a similar fashion. Preservative action is also dependent upon the solubility of the preservative in the aqueous phase, and its partition between water and oil phases. Micro- organisms grow in the water phase or at the water-oil interface. Therefore, the preservative should be in the water phase if it is to be effective. In an emul- sion, a preservative will partition itself depending on its relative solubility in
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)