RISK-BEARING SUBSTANCES IN COSMETICS 165 Table I. Inventory of eye make-up colours (numbers refer to colour index numbers) GREEN 77288 Chromic oxide 77289 Chromic oxide hydrate 75810 Copper chlorophyllene 77013 Ultramarine green -- Copper versenate -- Titanated mica (green) Synthetic organic colours and their lakes. e.g. 42170 44090 BLUE 77007 Ultramarine blue 77346 Cobalt aluminate 75510 Ferricferrocyanide -- Titanated mica (blue) Synthetic organic colours and their lakes. e.g. 42735 42090 Sudan blue II VIOLET 77007 Ultramarine violet 77745 Mna(PO4)a.7HaO 77742 Mn(NH4)PaO7 Synthetic organic colours and their lakes. e.g. 73385 YELLOW-ORANGE 77489 Iron oxide yellow 75300 Turmeric 75120 Anatto 75130 Carotene -- Titanated mica (gold) Synthetic organic colours and their lakes. e.g. 11920 19140 RED 75470 Carmine 77491 Iron oxide red 77007 Ultramarine red -- Titanated mica (red) Synthetic organic colours and their lakes. e.g. 12120 15850 45430 12150 15865 45170 12085 15880 45425 15630 BLACK 77499 Iron oxide black 77267 Bone black 77266 Carbon black -- Vegetable black -- Graphite BROWN 77492 Iron oxide brown -- Burnt sienna -- Burnt umber -- Caramel PEARL -- Guanine -- Titanated micas/talc -- BLOC1, or precipitated on mica 77480 Gold powder 77820 Silver powder 77400 Copper/bronze powder 77000 Aluminium powder WHITE -- Talc 77891 TiO 77947 ZnOa -- Zn carbonate 77005 Kaolin 77002 Aluminum oxide 77120 BaSOa 77713 MgCOa 77220 CaCO3 77231 Gypsum -- SiOa -- Tin oxide -- Stearates of Li, Mg, Ca, A1, Zn
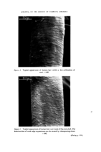
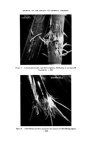
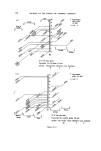
166 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Sulphuric acid conc. Sulphuric acid 4 M Hydrochloric acid 6 M NaOH 8 M Nitric acid 50•o Diphenylcarbazide 1 •o in ethanol Hydrogen peroxide 3•o Nessler reagent (K2HgI4) for ammonia Ammonium molybdate 0.5•o in conc. sulphuric acid Methanol Petroleum ether (40/60) Benzene Carbon tetrachloride Dimethylformamide Glycerol Borax Potassium bisulphate Potassium ferrocyanide Methods Additives should first be removed from the sample and the pigments isolated. There are three procedures to do this, which depend on the physicochemical properties of the sample. In these operations centrifuging is an important step, since the striated sediment will in general give visual information of the separated components. Removal of additives and isolation of the pigments. Procedure of removal of additives depends on the physicochemical properties of the sample: Hydrophilic (miscible with water): procedure A Lipophilic (immiscible with water): procedure B 'Wax cake' for instance mascaras: procedure C Procedure A. Mix 1 g sample with 40 ml water. Homogenize with blender or Ultra Turrax homogenizer. Centrifuge in 45 ml tube for 15 min at 3-4000 rev/min. N.B. The striated sediment will give valuable information, in particular, of which components the pigment-mixture consists. The analysis will proceed according to these assessed colour components of the dried sediment. The supernatant liquid might be coloured. If the colour can be adsorbed with Polyamide powder (procedure under Table III), the colour is in its water soluble form. If not the colour might be a suspension of small particles of dye-lakes, which is mainly present in the sediment. Procedure B. Mix 1 g sample with 20 ml petroleum ether. Heat gently on a water bath to defat the powder thoroughly. Decant the petroleum ether. Repeat the defatting procedure twice more. Dry the powder and suspend in 40 ml water. Proceed as under A. Procedure C. 'Wax cake' mascaras might contain chromic and iron oxides, and the different kinds of carbon blacks. For the identification of the carbon type
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)