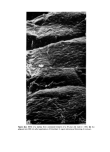
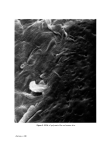
Figure 5. SEM of polymeric film on human skin. Facing p. 646
Water-resistant sunscreen preparation 647 Table IV. Human sweat study Sweat gland activity after 7-day use for 12 h per day Active sweat glands/cm • Sweat gland activity after 4 days continuous use Active sweat glands/cm • Sunscreen Untreated Sunscreen Untreated Saran Subject treated control Subject treated control occluded 1 144 149 7 106 98 78 2 119 122 8 81 80 50 3 99 96 9 85 81 30 4 103 103 10 105 91 17 5 86 80 11 77 82 23 6 47 56 12 89 96 23 No significant difference between sunscreen treated and untreated control by one-way analysis of variance. Saran occluded and untreated control are significantly different by one-way analysis of variance. Table V. Hairless mouse study Average grade Condition A B C 150 rain uvl 0'7 2-3 6 30 rain water immersion 1.6 5.6 7 + 150 rain uvl A, 3'3•o octyl dimethyl PABA plus film-former (lotion) B, 5'0•o PABA (hydroalcoholic lotion) C, untreated: 0-3 mild 3-5, moderate 6-7, severe. SAFETY TESTS Safety tests including Draize (204 subjects), maximization (twenty-eight subjects), cumulative irritation (twenty-seven subjects), phototoxicity (ten subjects), and photo- allergy (twenty-five subjects) established that the film-forming sunscreen product had little or no allergy or irritancy potential. Only slight irritation was noted on stripped skin in human volunteers with the film-forming formulation, whereas, other commercial preparations produced moderate to severe irritation (Table VI). Table VI. Human stripped skin irritation Mean Level of Sunscreen products irritation score irritation A 0.3 Slight B 1'4 Slight to moderate C 2.7 Moderate to severe D 1.2 Slight to moderate E 3-1 Moderate to severe Untreated control 0'0 None A, 3.3• octyl dimethyl PABA plus film-former (lotion) B, 3'0}/o amyl dimethyl PABA plus 3.0•o glyceryl PABA (lotion) C, 5.0• PABA (hydroalcoholic lotion) D, 10'0•o sulisobenzone (lotion) E, 2'5•o amyl dimethyl PABA (hydroalcoholic lotion).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)