DETERMINATION OF DIAZOLIDINYL UREA 181 method was not validated or attempted for all possible matrices that may be encoun- tered. ACKNOWLEDGMENTS The authors are grateful to Dr. William Rosen, Dr. John Merianos, Tim Parsons, and David Steinberg of Sutton Laboratories/ISP for providing the diazolidinyl urea, the consumer samples, and their expert advice and comments. Thanks are also owed to Matthew Benning and Paul Suszczynski of ISP for the HPLC analysis for parabens of some of the formulations. Permission to publish by ISP is gratefully acknowledged. REFERENCES (1) Germall © II Product Brochure (Sutton Laboratories, Chatham, NJ). (2) T. Nash, Colorimetric determination of formaldehyde under mild conditions, Biochem. J., 55, 416- 421 (1953). (3) E. Sawicki, T. R. Hauser, and S. McPherson, Spectrophotometric determination of formaldehyde and formaldehyde-releasing compounds with chromotropic acid, 6-amino-l-naphthol-3-sulfonic acid (J acid) and 6-anilino-1-naphthol-3-sulfonic acid (phenyl J acid), Anal. Chem., 34, 1460-1464 (1962). (4) R. L. Shriner, R. C. Fuson, D. Y. Curtin, and T. C. Morrill, The Systematic Identification of Organic Compounds, 6th ed. (John Wiley & Sons, New York, 1980). (5) H. Engelhardt and R. Klinkner, Determination of free formaldehyde in the presence of donators in cosmetics by HPLC and post-column derivation, Chromatographia, 20(9), 559-565 (1985). (6) W. R. Summers, Characterization of formaldehyde and formaldehyde-releasing preservatives by com- bined reversed-phase cation-exchange high-performance liquid chromatography with postcolumn de- rivatization using Nash's reagent, Anal. Chem., 62, 1397-1402 (1990). (7) A. R. Stack and H. M. Davis, Liquid chromatographic separation and fluorometric determination of quaternium-15 in cosmetics, J. Assoc. Off. Anal. Chem., 67(1), 13-15 (1984). (8) R. J. Geise, N. I. Machnicki, and R. I. Ianniello, Determination of allantoin in cosmetic products by capillary zone electrophoresis, Anal. Lett. 27(1), 183-194 (1994).
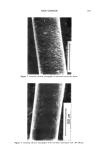
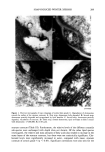
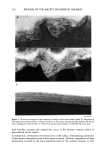
j. Soc. Cosmet. Chem., 45, 183-192 (July/August 1994) Damage of hair fibers as evaluated by an electrical capacitance technique HIRAKU ITO, YOICHIRO MURAOKA, and HARTWIG HOCKER, Heian Jogakuin (Saint Agnes') College, Nampeid•i, Takatsuki, Osaka 569, Japan (H.I., Y.M.), and Deutsches Wollforschungsinstitut, Veltmanplatz 8, Aachen D-S100, Germany (H.H. ). Received September 24, 1993. Synopsis An attempt was made to evaluate the damage of hair fibers using an electrical capacitance technique, and the results were compared with those obtained by using the Wilhelmy principle. To this end, two types of hair fiber samples were prepared. In one type, hair fibers were oxidized with dichloroisocyanuric acid (DCCA), and in the other, the fibers were physically rubbed with sandpaper. Water transport along the hair fiber bundle was examined with an apparatus using an electrical capacitance technique. The rate of water transport increased with the degree of damage. These results were consistent with the results on the contact angle measurements carried out with a Wilhelmy-type apparatus for recording the wetting force of a single hair fiber in water. Therefore, we found that an electrical capacitance technique is potentially useful as a convenient evaluation of the damage to hair fibers. INTRODUCTION Human hair fibers are treated repeatedly in many processes such as bleaching, dyeing, and permanent waving. As a result, hair fibers accumulate damaging effects. A method to diagnose the degree of damage of hair fibers would be very beneficial. However, no "convenient" method has yet been developed for evaluating the damage to hair fibers. As is well known, keratin fibers such as hair and wool have a so-called skin-core structure. The core, i.e., the cortex, is hydrophilic in nature due to the large number of polar groups contained in the polypeptide chains. On the other hand, the surface of keratin fibers is water-repellent, since the surface layer of cuticular cells (epicuticle) is hydrophobic in nature (1). Furthermore, there is a non-polar layer of fatty acid on the surface (2). When the keratin fiber is damaged by physical and/or chemical treatment, the structure of the fiber surface is modified. As a result, the surface property of the fiber is changed from a hydrophobic to a hydrophilic one. Therefore, the degree of fiber damage might be expected to correlate with the hydrophilicity of the fiber surface. Recently, we developed an apparatus using an electrical capacitance technique for eval- uating the water transport behavior along fiber bundles consisting of a few textile fibers 183
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)