PHOTOCHEMICAL REACTIONS 87 oxidizing agent. This intermediate, which may be either an excited dye- oxygen complex or singlet oxygen, then oxidizes the oxidizable substrate, ascorbic acid, with regeneration of the dye. After all of the oxygen in the system is consumed, an alternative reaction pathway involving electron transfer or hydrogen transfer from oxidizable substrate to the excited dye leads to photoreduction of the dye and loss of color. The alternative re- action pathways may be represented as (12-15): oxidizable h• D* D' substrate D • ,, • • Dred • So x I D'02 ] oxidizable substrate or , D + Sox D + singlet 02 D + 02 where D refers to dye in the ground or unexcited state, D * to dye in the singlet state, D' to dye in the triplet excited state, S to substrate, and the subscripts red and ox to reduced and oxidized species. Unquestionably, the oxidizable substrate, ascorbic acid in this case, is being oxidized during the period when the absorbance of the dye does not change and the reaction is therefore a photosensitized oxidation of ascorbic acid without destruction of the dye (Path A). In the absence of oxygen, the alternative reaction (Path B), involving electron or hydrogen transfer from the ascorbic acid to the excited dye triplet, leads to color loss of the dye and oxidation of ascorbic acid in a coupled oxidation- reduction reaction. Malachite green is a cationic dye and, although binding data are not available, the dye would be expected to bind extensively to an anionic micelle such as sodium decyl sulfate. Figure 4 illustrates the enhanced rate of photoreduction of malachite green by ascorbic acid in presence of sodium decyl sulfate, analogous to the effects of PMA and polysorbate 80. Malachite green would not be expected to bind so readily to a cationic micelle, but nevertheless 5% benzalkonium chloride enhances the photo- reduction approximately 2.3-fold. Data are summarized in Table I. Since these photochemical reactions proceed via a long-lived triplet state they can be quenched or retarded, in presence or absence of macro-
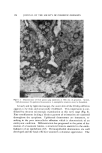
88 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS i I 1 I I I i 2_0 40 60 80 MINUTES Figure 4. Anaerobic photobleaching of malachite green in presence of 1 X 10-"M ascorbic acid. A, no additive B, 2% sodium decyl sulfate Table I Effect of Surface Active Agents on Rate of Photobleaching of Dyes- Surfactant k•urtactant/k 0 Malchite green-ascorbic acid (anaerobic photoreduction) None 1.0 1% Polysorbate 80 58 3% Polysorbate 80 200 5% Polysorbate 80 222 5% Triton X-100 8.6 2% Sodium decyl sulfate 7.8 5 % Benzalkonium chloride 2.3 Indigo carmine (photooxidation) None 1.0 2% Sodium decyl sulfate 0.51 5 % Benzalkonium chloride 0.20 • Rates determined from linear first-order plots of logarithm absorbance vs. time, 25øC The symbol k0 refers to rate of photobleaching without surfactants. molecules, by reagents which quench triplet states. Reagents for potential practical application in quenching excited states, and thus preventing photodecomposition, with particular application to cosmetic products were noted by Lewin (1). Another type of enhancement of photoreduction by polymers and surfactants has been observed even when there is no binding of the dye as determined by standard procedures such as solubility, equilibrium dialysis, partitioning between immiscible solvents, and ultraviolet and visible spectroscopy. Ribofiavin was found to exhibit an enhanced rate of aerobic photobleaching in presence of PVP and polysorbate 80, even
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)