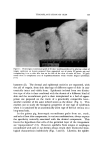
INTERMEDIATES FOR HAIR COLORS 143 4000 3000 2000 1500 CM -• 1000 900 800 700 100 : l! ' qil '- .... 7 i ": ",•:I. ]i 'i!:i' il ' iil 'i !: !t'T ,•'•I'ILI .i 11•i ' •'-'IH• 'H':i! : ..il. ' •00 ,•.f,,, '"- , -•--- •,•' • .... I • . .... • ..... •o • -.4:- J--:i : ,'• " " " ' ' •"• • •'•" :,-•' '4 .... •'::• -••• •: •'1 •/•M •-•'.:: •' •:'i ' .• • •-• : • • i 'ø 3 4 5 6 7 8 9 10 11 12 13 14 15 WAVELENGTH [MICRONS) Figure 1. Sludge from nitro-o-phenylenediamine Several factors may be responsible for the impnrities present in these nitro componnds inclnding the method of mannfactnring, the solnbility, and the ease of oxidation of the by-prodncts. Since nitro-o-phenylenedi- amine is prepared by the rednction of 2,4-dinitroaniline with alcoholic ammonium snlfide, some reducing agent may be present in the crnde prodnct. This was confirmed by a qnalitative test in which snlfnr di- oxide was given off on the addition of acid to the crnde product and a Parr Bomb determination which showed 0.28% of snlfnr in the crnde and none in the pnrified material. Another specnlation is that some 1,2,4-triaminobenzene may be formed by the rednction of both nitro gronps in the 2,4-dinitroaniline and that this prodnct is so readily oxi- dized that the 1% sodinto sulfite in the base solntion is insnfficient to prevent its oxidation on ageing. In the boiling test previonsly described, the addition of 10% of 1,2,4-triaminobenzene to the purified intermedi- ate resnlted in a color similar to that obtained with the crnde prodnct and an increase in the amount precipitated from 0.01 to 0.77%. A1- thongh the strnctnre of the black slndge, removed dnring recrystalliza- tion, is beyond the scope of this paper, its high melting point (above 335øC) indicates a complex structnre. Nitro-p-phenylenediarnine A method of preparing nitro-p-phenylenediamine is described in a recent patent (4). In this process, 2-nitro-l,4-dicarboxylic acid diamide is treated with sodinto hypochlorite which converts it to 2-nitro-l,4-di- carboxylic acid n-chloramide. Upon heating, carbon dioxide and chlo- rine are split off forming nitro-p-phenylenediamine. It is pnrified by re- crystallization from water. In more recently issned patents (5-7), a method is described for preparing nitro-p-phenylenediamine which con- sists of the nitration of p-fiuoroaniline nnder anhydrons conditions to
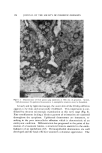
144 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS form 4-fluoro-3-nitroaniline, followed by reaction with ammonia to re- place the fluoro group. The solubility of nitro-p-phenylenediamine at 25øC in aqueous solution at a pH of 9.7 is 0.23% (1). Comparable analy- ses of this intermediate before and after recrystallization are given in Table II. Table II Analyses of Nitro-p-phenylenediamine Purified Raw Purity, % 98.1 90.1 Ash, % Trace 0.20 Iron, ppm Trace 363 Melting point (Fisher-Johns), øC 143.6 134.0 In the ageing test previously described, precipitation in the solution containing the crude product started after 11 days, both at room tempera- ture and at 50øC, while no precipitate appeared in bottles containing the purified material after ageing for 4 weeks. The infrared spectrograms for nitro-p-phenylenediamine are shown in Figs. 2 and 3. Certain reversals in the peaks, especially in the range 4000 30'00 2000 1500 CM" 1000 900 800 700 10C 100 __8C B0 •6C 60 •4C 40 Z •- 2C 3 4 5 6 7 8 9 10 11 12 13 14 WAVELENGTH JMICRONS) Figure 2. Crude nitro-p-phenylcncdiamine 4000 30'00 2000 1500 C• -I 1000 900 800 700 100 100 •.80 80 •o • 40 j.,o , •- 20 210) 3 4 5 6 7 8 9 10 11 12 13 14 1 WAVELENGTH (MICRONS) Figure 3. Purified nitro-p-phenylenediamine
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)