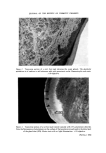
JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 5 13 Et O (nominal) Figure 3. Thin-layer chromatogram of oleyl alcohol ethoxylates. Plate silica gel G. Solvent methyl ethyl ketone saturated with water. Chromogenic reagent iodine vapour. Left, oleyl alcohol ethoxylate (5 Et 0 units per mole, nominal). Right, oleyl alcohol ethoxylate (13 Et 0 units per mole, nominal). (Facbtg p. 690)
EYE IRRITATION 691 more difficult to decide whether to keep the product on the market. The hairdressing under discussion gave rise to reports of visual disturbances in about one in 8 500 users such a level even for a relatively moderate adverse reaction, certainly contra-indicated extension from test market to a na- tional launch. A surprising feature was that eye irritancy testing on laboratory animals failed completely to indicate the likelihood of adverse effects in man. How- ever, corneal pitting is not generally regarded as an important criterion in eye irritancy testing. In the present instance, the most disturbing symptom took the form of visual blurring and ophthalmologists examining some complainants noted the occurrence of pitting of the corneal epithelium (in one case described as a superficial punctate keratitis) it seems reasonable to believe that such pitting (possibly in conjunction with other changes) could interfere with visual acuity sufficiently to produce a considerable degree of blurring. Fortunately, the effect did not imply serious or irreversi- ble damage, but the degree of distress was undoubtedly sufficient to suggest that corneal pitting merits closer attention in predictive testing. Even though the failure to predict human eye irritancy from 'conven- tional' laboratory testing could subsequently be ascribed partly to inatten- tion to transient corneal pitting, the causation of this response was not obvious. For example, the complete formulation did not produce any marked adverse effect when instilled into the eyes of human volunteers nor was there generally any response to the formulation in the eyes of laboratory animals. The time between washing the hair and exposure to rain was quite variable in the complaint cases and it therefore seemed unlikely that a slow chemical degradation process was responsible. After applying an emulsified product to the skin or hair, physical changes are usually obvious the residue after evaporation seldom bears a close resemblance to the original formulation. When the transparent micro-emulsion discussed here had aged on the scalp, the addition of water yielded an eluate with a milky appearance quite unlike the original preparation furthermore there was no means of knowing the actual concentration of the eluate which reached the com- plainants' eyes. Neither the ethoxylate nor any other ingredient behaved as an eye irritant when it was present within the interfacial film of the emulsion. Un- like the other constituents, however, an aqueous solution of the ethoxylate did exhibit a degree of eye irritancy approximating in some respects to the irritancy experienced by complainants. Since the original formulation was
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)