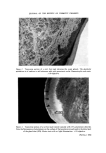
SURFACE FORCES IN THE DEPOSITION OF SMALL PARTICLES 711 (a) Figure 1. Particle/plate contacts. (a) The action of surface tension on a platy particle, and (b) on a spherical particle (c) represents the small contact points between rough surfaces. truly microscopic particles it becomes far more important than the weight. However, the point of interest is what happens after the liquid evapor- ates does the particle remain attached, or does it fall off--if not under its own weight, then perhaps with a small applied mechanical force? This question is dearly about adhesion that turns out to be a rather different phenomenon from the process of attachment of particles, which will be referred to as deposition. The one is not simply the reverse of the other because once deposition has occurred, which is determined by relatively long-range forces, then close-range forces come into action and these generally produce changes in the system--often slow changes, so that strength of adhesion changes with time. On reflection, it is clear that although the sphere/plate model may be
712 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS useful for the study of deposition phenomena, it is unlikely to be adequate for adhesion studies. Firstly, the real particles and substrates are not mathematically smooth consequently, their zone of contact is not a single point, but two or three spots of sub-microscopic area (Fig. lc). Since these spots must be determined by chance (as a stone touches a rock), it is not surprising that adhesion experiments always reveal a wide range of strengths of adhesion for a nominally uniform population of particles. This is fundamentally because the short-range forces are more powerful than long- range ones, and therefore adhesion is sensitive to the fine detail of the con- figuration of the bodies near the points of contact. Secondly, the pressure acting on the points of contact may be large enough to cause some flattening of liquid droplets, elastic deformation of hard solids and plastic deformation of soft ones. The effect is not easy to measure, but it may have a significant effect on the adhesion. Thirdly, if the particles are even slightly soluble in the liquid environ- ment from which they are deposited, the thermodynamic effect of pressure on the point of contact can significantly increase the local solubility, causing the projection to dissolve away from the point and recrystallize in the vicinity. Thus, narrow contact spots eventually become transformed into wide supporting pillars, and the adhesion strength increases with time. This effect has been well demonstrated by Uno and Tanaka (29) with crystals of chloramphenicol on glass. (The effect is entirely distinct from recrystalliza- tion of contacts as a result of temperature fluctuations or evaporation.) Adhesion is seen to be a decidedly more complex problem than deposi- tion. It has been reviewed in considerable detail by Krupp (19) and will be only briefly reviewed below. In the absence of bridge formation, adhesion of particles to smooth solids is almost entirely confined to the truly micro- scopic or sub-optical range, and consequently direct measurement of the force of adhesion is not easy. The oldest method, used by yon Buzagh (26), was to measure either the angle of tilt of the plate required to start the particles moving or the propor- tion of particles remaining attached after the plate had been slowly inverted. This type of experiment was carried out mainly with small quartz particles and aqueous electrolytes, the particles being observed through a low-power microscope. Adhesion was found to be most marked with particles of around 3 gm diameter and was favoured by addition of electrolytes. Although this method is simple and reveals qualitative effects, its quantitative value is limited. Sliding angles with large particles are indicative of friction to move- ment along the plane, rather than the normal force for detachment.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)