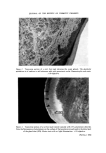
SURFACE FORCES IN THE DEPOSITION OF SMALL PARTICLES 717 Material Medium Figure 3. Model employed in heterocoagulation theory. Heterocoagulation The theory of heterocoagulation, allowing for differences of potential of the two materials (q• and q•2) and mutual Hamaker-van der Waals con- stant A•a2, for attraction between materials 1 and 2 while immersed in medium 3, was first given by Derjaguin (1) (cf. 2, 3, 5). A useful practical approximation for heterocoagulation of unlike particles of unequal size, given by Hogg, Healy and Fuerstenau (3), takes the form Vtotal = VR 'Jr' V A with VR double-layer repulsion energy and VA the van der Waals attraction being given by VR=F 7 2+•+•.ln 1-exp(-KH)l + (½•2++•)ln[1-exp(-2KH)] a VA=F.-- 6H
718 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS F is the form factor axad(ax + a•) and • is the Debye-Hfickel reciprocal length, which is related to the ionic strength of the medium. This theory has been used to predict the conditions for heterocoagula- tion, the influence of particle size (4) and the possibility of selective coagula- tion in mixed colloidal dispersions (7). It should also apply to deposition, with F= ax. In employing the theory, it is usual to assume that the surface potentials, ½x and 4e•., can be replaced by the practical zeta potentials of the materials. Generally, there is no alternative. Some uncertainty exists as to whether colloidal interactions occur under conditions of constant surface potential or constant charge. In classical D.L.V.O. literature, in which constant potential was assumed, it was claimed that there would not be a great difference between the two but, in the case of heterocoagulation, the constant potential assumption leads to an attraction force at close interaction between surfaces of high and low potential of the same sign. The physical origin of this force is clear enough, but there is no experimental evidence for it. Some authors now calculate on both hypotheses, arguing that the truth lies somewhere between the two. Computations show appreciable differences in stability factors (7). It may be possible to employ the rotating disc technique to settle this question. Van der Waals forces Until recently, the attraction energy was always calculated by the Hamaker method which, following London, de Boer and others, assumed that the attractive force between colloidal bodies arises from London dis- persion forces and that these are substantially additive. Although it was recognized that numerical estimations of the interaction coefficient from optical data etc. were necessarily rather approximate (9), (15), for a single substance (in vacuum) the Hamaker constant, Axx, could be calculated within an accuracy of, perhaps, -t-50•o although Visser's recent review reveals far larger discrepancies in many cases (14). The interaction between dissimilar particles is believed to follow approximately a geometrical law.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)