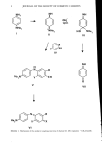
OXIDATIVE HAIR DYEING 7 -2 -3 -5 O Ot I I • I I 8 10 12 pH Figure 1. Variation of log kobs with pH for the loss in water at 30øC of 2-aminoindamine formed from N,N-bis-(2-hydroxyethyl)-p-phenylenediamine and m-toluenediamine. Theoretical line shown with experi- mental points superimposed. COLOR ON HAIR AND DYEOUT STABILITY Piedmont hair swatches were dyed for 30 min using 1% of the disubstituted diamine and 1% of each of the specified couplers in a commercial dye base and an equal volume of 6% hydrogen peroxide solution. Swatches were then stored for 68 weeks at 0% and
8 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table III Rate Constants (k2) for Cyclization of Aminoindamines (VIII, X = NH) at 67 ø and Spectral Data [h.m• (log ½)] for the 2,8-diaminophenazine Products Indamine Phenazine R R' R • Cation pKa Neutral 104k2 (s- •) CH2CH2OH CH2CH2OH H 534 (4.50) 6.8 460 (4.19) 2.3 CH2CH2OH CH2CH2OH CH 3 535 (4.56) 6.6 463 (4.18) 1.4 a CH:CH:OH CH:CH:OH OCH 3 533 (4.21) 6.4 445 (4.04) 0.2 CH2CH:OH CH2CH:OH C1 530 (4.43) 6.2 460 (4.14) 10.2 H H CH 3 506 (4.30) 6.5 438 (4.13) 1.5 CH 3 CH 3 CH 3 518 (4.50) 6.3 452 (4.23) 4.0 H CH2CH2OH CH 3 520 (4.55) 6.5 445 (4.34) 6.9 H CH 3 CH 3 515 (4.61) 6.7 445 (4.40) 10.7 a From reactions at different temperatures, E a was evaluated as 21.9 Kcals/mole. Table IV Stability of Dyeings on Hair From N,N-bis-(2-hydroxyethyl)-p-phenylenediamine and Various Couplers Coupler Initial Color After 68 weeks at 80% RH p-diamine alone Green/gray Tan m-toluenediamine Peacock blue Blue/gray m-aminophenol Violet Brown/gray 5-amino-0-cresol Violet Light brown Resorcinol Light brown Light brown 80% relative humidities. The color before storage is shown in Table IV. After storage at 0% RH, there had been very little color change. For example, Hunter Tristimulus readings on a swatch using 5-amino-0-cresol as coupler were L = 22.7, a = 7.0, and b = - 10.8 initially, and L = 22.4, a = 7.6, and b = -8.4 after storage. In con- trast, however, swatches stored at 80% RH had become lighter with the development of brownish shades (L = 35.9, a = 1.8, and b = 7.1 on above example). Colors developed from the other couplers are shown in the Table. These dyeouts are generally more stable than those from either p-phenylenediamine or p-aminophenol, and the fading is in a direction similar to that observed with p-phenylenediamine. Clearly, therefore, these results correlate with the solution chemistry of the indo dyes. REFERENCES (1) D. J. Palling, K. C. Brown, and J. F. Corbett, Benzoquinone imines, Part 18.' Kinetics and mecha- nism of oxidative coupling reactions involving N,N-bis-(2-hydroxyethyl-p-phenylene-diamine, J. Chem. Soc., Perkin II, 65-68 (1986). (2) J. F. Corbett, S. Pohl, and I. Rodriguez, Benzoquinone imines, Part XII: Reactions of 2-aminoinda- mines in aqueous solution, J. Chem. Soc., Perkin II, 728-734 (1975). (3) K. C. Brown and J. F. Corbett, The role of meta difunctional benzene derivatives in oxidative hair dyeing.. II. Reactions with p-aminophenols, J. Soc. Cosmet. Chem., 30, 191-211 (1979). (4) J. F. Corbett, Chemistry of hair colorant processes--Science as an aid to formulation and develop- ment, J. Soc. Cosmet. Chem., 35, 297-310 (1984).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)