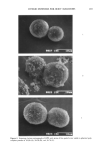
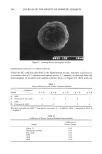
AMPHIPHILIC ASSOCIATION STRUCTURES 169 Figure 13. An isotropic solution (left) is dark when viewed between crossed polarizers. A liquid crystal, on the other hand, is anisotropic (and hence birefringent) and radiant (right). The microemulsions _per se are in equilibrium with a liquid crystal (Figures 2 and 8). Hence, compositions inside the microemulsion region will dissolve a liquid crystal when brought in contact with it. The ramifications for the interaction between micro- emulsions and the stratum corneum are obvious a microemulsion in intact form will dissolve the ordered stratum lipid structure to form a liquid. With the lipids in a disordered liquid form, the barrier to water is lost and--what is more important--the barrier to transdermal transport inwards is also damaged. It is a well known fact that facilitated transdermal transport of foreign substances in the majority of cases leads to irritation. This kind of irritation may be called structure-induced irritation because its main cause is the enhanced permeability due to changed stratum corneum structure. However, the water in the microemulsion does evaporate in 10-20 minutes, and the actual influence by the microemulsion on the skin structure is determined by the struc- ture of the residue after the water is removed. If the remains on the skin are an oil solution of the surfactant (Figure 10), a disordering of the stratum corneum lipid structure with the consequence of irritation is a possi- bility. If, on the other hand, the final structure is a liquid crystal (Figure 10), the risk of the structure-induced irritation does not exist, but nonirritating surfactants for the microemulsions must be chosen. The emulsion system (Figure 5) has identical condi- tions. The vesicles or liposome solutions, on the other hand, automatically transform to a lameliar liquid crystal after evaporation of the water. Compositions of this kind should be considered safe from structural considerations, but the final lameliar structure is, of course, no protection against irritating action of individual components. It should be emphasized that the knowledge about the stratum corneum structure and about the association structures of ointment remainders makes it possible to give a precise evaluation of the influences on the stratum corneum structure. SUMMARY The amphiphilic association structures were shown to be involved in all parts of the cosmetic process, from the formulation efforts over the changes taking place when the
170 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS composition was applied to the skin to the changes of stratum corneum lipid structure brought forward by its influence on the stratum corneum lipids. ACKNOWLEDGMENTS This research was supported financially by Colgate-Palmolive Central Research Labora- tories within the CAMP program at Clarkson University. REFERENCES (1) Cosmeticology, J. B. Wilkinson and R. J. More, Eds. (Chemical Publishing, New York, 1981). (2) Cosmetic Science, M. M. Breuer, Ed. (Academic Press, London, 1980). (3) M. M. Breuer, "Cosmetic Emulsions" in Encyclopedia of Emulsion Technology, P. Becher, Ed. (Marcel Dekker, New York, 1985), Vol. 2, pp. 385-424. (4) J. N. Israelachvili, D. J. Mitchell, and B. W. Ninham, Theory of self-assembly of hydrocarbon, J. Chem. Soc. Faraday Trans. II, 72, 1525-1567 (1976). (5) P. Becher, Emulsions: Theory and Practice, 2nd ed. (Reinhold, New York, 1968). (6) Emulsion Science, P. Sherman, Ed. (Academic Press, London, 1968). (7) K. Shinoda, The correlation between the dissolution state of ionic surfactant and the type of disper- sion stabilized with the surfactant,J. Colloid Interface Sci. 24, 4-9 (1967). (8) T. J. Lin, Low energy emulsification. 1. Principles and applications, J. Soc. Cosmet. Chem., 29, 117-125 (1987). (9) S. E. Friberg, L. Mandell, and M. Larrson, Mesomorphous phases, a factor of importance for the properties of emulsions, J. Colloid Interface Sci., 29, 155-156 (1969). (10) Food Emulsions, S. E. Friberg, Ed. (Marcel Dekker, New York, 1976). (11) H. Sagitani, Making homogenous and fine droplet o/w emulsions using nonionic surfactants, J. Am. Oil Chem. Soc., 58, 738-743 (1981). (12) T. Suzuki, H. Tsutsumi, and A. Ishido, Secondary droplet emulsion: Mechanism and effects of liquid crystal formation in o/w emulsion, J. Disp. Sci. Techn., 5, 119-141 (1984). (13) Microemulsions.' Structure and Dynamics, S. E. Friberg and P. Bathorel, Eds. (CRC Press, 1987). (14) K. Shinoda and B. Lindman, Organized surfactant systems: Microemulsions, Langmuir, 3, 135-149 (1987). (15) S. E. Friberg, "Stabilization of Inverse Micelles by Nonionic Surfactants," in Interfacial Phenomena in Apolar Media, H.-F. Eicke and G. D. Parfitt, Eds. (Marcel Dekker, New York, 1987). (16) H.oF. Eicke and H. Christen, Nucleation process of micelle formation in apolar solvents, J. Colloid Interface Sci., 48, 281-287 (1974). (17) B. H. Robinson, C. Toprakcioglu, and J. C. Dore, Small angle neutron scattering studies of micro- emulsions stabilized by aerosol-OT, J. Chem. Soc. Faraday Trans., 80, 13-27 (1984). (18) S. E. Friberg and I. Buraczewska, Microemulsions in the water-potassium oleate-benzene system, Progr. Colloid Polym. Sci., 63, 1-9 (1978). (19) E. Sj6blom and S. E. Friberg, Light-scattering and electron microscopy determinations of association structures in w/o microemulsions, J. Colloid Interface Sci., 67, 16 (1978). (20) D. G. Rance and S. E. Friberg, Micellar solutions versus microemulsions, J. Colloid Interface Sci., 60, 207-209 (1977). (21) S. Wahlgren, A.-L. Lindstr6m, and S. E. Friberg, Liquid crystals as a potential ointment vehicle, J. Pharm. Sci., 73, 1484-1486 (1984). (22) A.M. Kligman in Biology of the Stratum Corneum in Epidermis, W. Montagna, Ed. (Academic Press, New York, 1964), pp. 421-446. (23) P.M. Elias, B. E. Brown, P. T. Fritsch, R. J. Gorke, G. M. Goay, and R. J. White, Localization and composition of lipids in neonatal mouse stratum granulosum and stratum corneum, J. Invest. Dermatol., 73, 339-348 (1979). (24) S. E. Friberg and D. W. Osborne, Small angle x-ray diffraction patterns of stratum corneum and a model structure for its lipids, J. Disp. Sci. Techn., 6, 485-495 (1985).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)