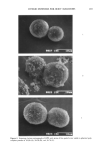
UV-INDUCED SKIN TUMORS 189 SUBJECTS Four normal healthy Caucasians between the ages of 55 and 81 with Fitzpatrick skin types of I, II, or III (33) and a past history of chronic solar exposure were included in the study. Subjects were selected on the basis of severe chronic photodamage to the dorsal neck, outer arm, or preauricular area of the face. Little photodamage existed on the upper back, buttock, or inner arm. None of the subjects showed signs nor had a history of skin cancer. Skin sites were graded subjectively on a 0-5 scale for photo- damage by an experienced grader. Extensively photodamaged skin (grade 5) was yel- lowish, wrinkled, and had poor elastic mechanical properties (determined by its resil- iency after extension). All subjects had refrained from taking antihistamines, antibi- otics, anti-inflammatory agents, and/or vitamins for at least two weeks prior to the study. None of the subjects had recently been exposed to either natural or artificial sunlight. An internal review board (Hill Top Research, Cincinnati, OH) approved all protocols used in this work, and all volunteers signed informed consent statements prior to the study. ANIMALS Female albino hairless mice (Skh:HR-1 from Charles River, Portage, MI) were used at 10- 15 weeks of age. Mice were housed five to a cage in a room with controlled temper- ature and humidity and with a 12-hour light/darkness cycle. Mice were then individu- ally identified by ink-colored tail markings and cage number throughout the course of the experiments (e.g., cage 1, red cage 1, green, etc.). They were given a standard diet and water ad libitum. UV IRRADIATION The UVR sources were either a solar simulator (Kratos, Ramsey, NJ) equipped with a 1000-W xenon arc lamp and dichroic filter or a bank of four Westinghouse FS-40 UVB sunlamps. Spectral emission of each UVR source was measured with an International Light 791 radiometer (Newburyport, MA) as previously described (18). For chronic UVB irradiation, mice were irradiated (3 X/week) in individual wire cage chambers (7.5 X 7.5 X 7.5 cm) in which their backs were 35 cm from the fluorescent tubes. UVB irradiance at this distance was 0.23 mW/cm 2 as measured with an International Light 700 radiometer total exposure dose was 55 mJ/cm 2 for each irradiation [•-0.8 mouse minimum erythemal dose (MED)]. For chronic simulated solar radiation (SSR), mice were individually irradiated (3 X/week) with their backs 10 cm from the dichroic filter, as previously described (28). SSR irradiance at this distance was 18 mW/cm2 total exposure dose was initially 540 mJ/cm 2 (•0.8 x MED) and was increased ap- proximately 30% every two weeks until a total daily dose of 900 mJ/cm 2 was reached, after which the dose remained constant for each irradiation. TOPICAL TREATMENTS There were 15-20 mice in each treatment group. Treatments [0.05 ml sunscreen 0.1 rnl 5% o•-tocopherol or 0.1 ml 5% iron chelators (w/v)] were applied topically to the
190 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS dorsal back skin using a 1-cc syringe and spread evenly with a glass rod (sunscreen) or a Pipetman © (Rainin Instrument Co., Woburn, MA) and spread evenly with the flat side of the pipet tip (ot-tocopherol and iron chelators). When groups were treated and irra- diated, treatments were applied two hours prior to UVR exposure except for sunscreen treatments, which were given immediately prior to exposure. For combination che- lator-plus-sunscreen treatments, the chelator was applied two hours before UVR expo- sure, followed by the sunscreen applied immediately before exposure. After treatment, mice were returned to their cages without restraint until irradiation. EXPERIMENTAL PROTOCOL The protocol used in this study is shown in Figure 1. Groups of hairless mice were subjected to a UVR exposure regimen (3 X/week) for 15 weeks without topical treat- ment. The UVR sources were either a solar simulator or UVB fluorescent tubes. After the UVR pre-exposure period, topical treatments were begun (3 x/week) with the pho- toprotective agents. During the treatment period, some groups of mice were continued on the UVR exposure regimen (continued UVR), while others were treated without further irradiation (stop UVR). After the treatment period (12 or 20 weeks), total tumor area and basal epidermal ODC activity were measured. This protocol mimics the probable human situation of some level of solar damage to the skin before the first use of photoprotective product. The purpose of either continuing or discontinuing irradiation after starting treatment is to mimic the human situation of either continued further sun exposure or avoidance of further sun exposure. TUMOR QUANTIFICATION Skin lesions were diagnosed as tumors if they were round, red, raised, and greater than 1 mm in diameter. Total tumor area per animal was accurately quantified from photo- graphic transparencies of the backs of the animals by using a computerized digitizing tablet as previously described (28). Continue UVR Exposure I Stop UVR , Exposure ' No Treatment Photoprotective Period Treatment Period I I 5 10 15 20 25 30 35 Time (weeks) Figure 1. UV irradiation and treatment protocol.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)