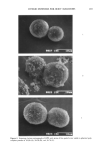
180 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table I Compounds Tested for Inhibition of Aryl Sulfatase and beta-Glucuronidase Enzyme inhibition For beta G: Cu + + Zn + + SPORIX D-glucaro-A-lactone EDTA NTA O-phenanthroline Citrate Sodium sulfate Gluconate Mannose Fucose Galactosamine Pullulan For AS: Cu + + Zn + + SPORIX hexametaphosphate Orthophosphate EDTA NTA quantitative assays were studied quantitatively in a fluorescence assay. As in the semi- quantitative tests, the progress of the reactions was indicated by the development of fluorescent product 4-MU from the non-fluorescent substrates, 4-MUG and 4-MUS. The data was recorded as fluorescence emission intensity as a function of time. A repre- sentative example of the results of these assays is given in Figure 5, which shows the effect of glucarolactone concentration upon beta-G activity. Similar plots were con- structed for each inhibitor. At each concentration, the beginning slope indicated initial reaction rate. These rates were then expressed as a fraction of the uninhibited reaction rate and plotted against the log of the inhibitor concentration (Figures 6 and 7). For beta-G (Figure 6) the glucarolactone was the most effective inhibitor (1-10 range). The divalent cations, Zn + + and Cu ++, were effective at approximately 10-100 IxM. The sequestering agents EDTA and phenanthroline were significantly less effective, providing inhibition at millimolar levels. For AS, the Cu + + ion was most effective (0.1-10 IxM range). Zn + + and phosphate were effective at 10- 100 I. tM. Most microbial AS belongs to the Type I class, which is not inhibited significantly by sulfate or phosphate, but the AS of Aerobacter aerogenes (our enzyme) is somewhat more sensitive than that of other bacteria (30). This property is apparent in our study, where phosphate is found to be about as effective as zinc cation. These studies show that at least some bacterial beta-G and AS can be inhibited by Zn, Cu, chelating agents, or glucarolactone.
AXILLARY MALODOR 181 -lO i i i i i 10 20 30 40 50 60 lime, min. Figure 5. Inhibition of beta-glucuronidase by glucarolactone. 0.000 mM 0.004 mM 0.010 mM 0.020 mM 0.040 mM 0.130 mM 0.200 mM 120 100 t •{ so • 40 ! ! ._ Glucarolactone Zn ++ Cu ++ o-Phenanthroline EDTA log inhibitor concentration, uM Figure 6. Inhibition of beta-glucuronidase: A decrease in relative reaction velocity with increasing con- centration of five inhibitors.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)