744 JOURNAL OF COSMETIC SCIENCE Internal standard. Ethoxybenzene (0.1 mg/mL in dichloromethane) was used as an internal standard for the quantification of the FRMs. Sample preparation. Six tresses (0.5 g/20 cm long) from each group were washed for 1 min with the shampoo formulation containing the FRMs (50 mg/0.5 g of hair) and rinsed under tap water (30 ± 3°C) for 30 s. Three of these tresses (T0 samples) were immediately placed in 10 mL glass flasks with lids containing 2 mL of dichloromethane. The other three tresses (T24 h sample) were subjected to the same process after drying under controlled conditions (22 ± 2°C and 50 ± 5% RH) for 24 hours. After 24 hours of extraction, the hair was removed from the flasks, and 100 μL of the internal standard solution was added to each flask. The samples were manually homogenized, dried with anhydrous sodium sulfate (Nuclear, CAQ, BRL) and filtered through a 0.2 μm PTFE filter (ValuPrep™ 25 mm, PALL Life Sciences, USA) to 3 mL chromatography vials. Response factor. Six tresses (0.5 g/17–20 cm long) from each group were washed with the shampoo formulation without the FRMs (50 mg/0.5 g hair) for 1 min and rinsed under tap water (30 ± 3°C) for 30 s. Each tress was placed in a 10 mL glass flask with a lid, with 2 mL of dichloromethane, and 100 μL of a solution of the five raw materials in dichloromethane at 0.1 mg/mL each. This procedure was performed immediately after washing (T0 samples) or after drying under controlled conditions (22 ± 2°C and 50 ± 5% RH) for 24 hours (T24 h samples). After 24 hours of extraction, the hair was removed from the flasks, and 100 μL of the internal standard solution was added to each flask. The samples were manually homogenized, dried with anhydrous sodium sulfate (Nuclear, CAQ, BRL), and filtered through a 0.2 μm PTFE filter (ValuPrep™ 25 mm, PALL Life Sciences, USA) to 3 mL chromatography vials. GC-MS analysis. The samples were injected in a gas chromatograph (6890N, Agilent Technologies, USA) with an automatic injector (Multi-Purpose Sampler 2, Gerstel, USA) coupled to a mass detector (Mass Selective Detector 5973, MSD 5973 Agilent Technologies, USA). A ZB-1MS column (60 m × 0.25 μm × 250 μm) was used for component separation. The injection mode was set to the splitless mode. The oven temperature program was adjusted to heat up to 50°C, hold for 8 min at this temperature, and then heat up to 240°C. The samples were analyzed in the selected ion monitoring mode after choosing three appropriate mass/load ratios for each raw material at the time of the assembly of the method in the equipment software (ChemStation, Agilent Technologies, USA). Table II Shampoo Formulation Component (INCI name) Concentration (%) Water q.s.100.00 Disodium EDTA 0.05 Polyquaternium-10 0.20 Glycerin 1.00 Sodium Laureth Sulfate 29.63 Cocamidopropyl Betain 8.00 1,2-Hexanediol, Caprylyl Glycol 0.80 Trideceth-9, PEG-5 Isononanoate, Water 1.00 PEG-150 Distearate 0.50 Cocamide DEA 1.50 Citric Acid q.s. pH 5.5–6.5 Sodium Chloride 0.15
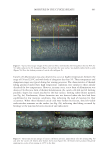
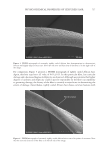
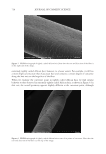
745 INFLUENCE OF ETHNICITY AND DAMAGE LEVELS Substantivity calculation. FRM substantivity was calculated using Equation 1. Substantivity(%) Std weight FRM peak area Std peak area RF * * = * *100 FRMweight Equation 1 RESULTS AND DISCUSSION HAIR DAMAGE ASSESSMENT The results obtained for the damage characterization of hair tresses are summarized in Table III. In the comparison among the spectra obtained for virgin hair, the most important differences were observed in hair tresses that were straightened plus bleached for all the evaluated ethnicities. For these samples, hair oxidation caused by the oxidative agents present in this type of treatment was evidenced by the increased intensity of the band centered at 1,043 cm−1 (assigned to −S=O symmetric stretch), and the widening of the band at 1,177–1,233 cm−1 (characteristic of sulfonate and thiosulfate groups and portions of cysteic acid) (9–11). From the stress–strain curves obtained in the tensile test, we evaluated two important parameters: elastic modulus and break stress. In general, straightening followed by thermal damage was the process that generated the most severe changes in hair structure. However, all the evaluated damage processes resulted in an increase in the hair elastic modulus and a decrease in the break stress, thereby increasing its stiffness and fragility in comparison to virgin hair. For all evaluated ethnicities, the straightened and thermally damaged hair lost the highest amount of protein among the damage groups. As expected, virgin hair showed extremely low values of protein loss, which was statistically smaller than those of the other groups in all three ethnicities. Table III Summary of Damage Characterization Results Damage group Ethnicity FTIR–ATR Tensile Protein loss (mg/g of hair) Swelling (%) elastic modulus (MPa) Break stress (MPa) Straightened African No alteration +6%*** −7%*** +740%*** +38%*** Asian No alteration +4%* −3%* +1,059%*** +39%*** Caucasian No alteration +12%*** −5%*** +967%*** +35%** Straightened + thermally damaged African No alteration +31%*** −12%*** +1,851%*** −19%* Asian No alteration +29%*** −6%*** +2,651%*** −43%*** Caucasian No alteration +37%*** −2% +2,500%*** −35%*** Straightened + bleached African Oxidation +28%*** −4%* +920%*** +120%*** Asian Oxidation +27%*** −4%** +1,068%*** +161%*** Caucasian Oxidation +30%*** −3%* +736%*** +99%*** ***p 0.001, **p 0.01, and *p 0.05 versus virgin hair from same ethnicity.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)