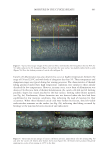
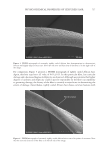
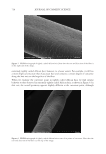
708 JOURNAL OF COSMETIC SCIENCE exposure (8). Specifically, a marker peptide at m/z 1,278 was identified as a fragment of the S100A3 protein known to be involved in cuticle cell adhesion (32). Hair treated with four of high activity tea extracts was exposed to UV radiation and compared to hair with no treatment exposed to UV radiation and hair with no UV exposure. After UV exposure the protein biomarker at m/z 1,278 was extracted in water and quantified by MALDI-TOF MS. A significant increase in biomarker peak intensity was measured for hair that had been pretreated with tea extracts, illustrating a protective benefit. The magnitude of this benefit was correlated to the concentration-adjusted ORAC score (R2=0.98) CONCLUSION The UV oxidation of proteins entails complex chemistry involving reactive oxygen species, and tracking this chemistry in hair is extremely challenging. Our approach has been to use a model colloidal system incorporating tyrosine, a key amino acid involved in UV-induced protein damage. Using this colloidal system, we identified the roles of oxygen, reactive oxygen species, and redox metals such as iron in the oxidation of tyrosine. Also demonstrated with this model system was the ability to decrease oxidation through the addition of antioxidants such as BHT. Experiments on hair were performed with selected botanical extracts of tea (C sinensis) that contain polyphenols such as catechins that are efficient radical scavengers as demonstrated by high ORAC scores. The efficacy of tea extracts was found to correlate with the level of tea compounds and with the levels of catechins and procyanidins, highlighting the importance of understanding botanical extract phytochemistry when assessing antioxidant performance. The highest-efficacy tea extracts were shown to prevent UV damage to hair proteins as measured by a reduction in the formation of a protein biomarker (m/z 1,278). ACKNOWLEDGEMENTS The authors thank The Procter & Gamble Company for funding support of this work. REFERENCES (1) T. Evans and R. R. Wickett, Eds., Practical Modern Hair Science (Allured Publishing Corporation, Carol Stream, IL, 2012). (2) C. M. Pande and J. Jachowicz, Hair photodamage: measurement and prevention, J. Soc. Cosmet. Chem., 44, 109–122 (1993). (3) E. Hoting and M. Zimmerman, Photochemical alteration in human hair. Part III: investigation of internal lipids, J. Soc. Cosmet. Chem., 47(4), 201–211 (1996). (4) W. Korytowski, B. Pilas, T. Sarna, and B. Kalyanaraman, Photoinduced generation of hydrogen peroxide and hydroxyl radicals in melanins, Photochem. Photobiol., 45(2), 185–190 (1987). (5) J. M. Dyer, S. D. Bringans, and W. G. Bryson, Determination of photo-oxidation products within photoyellowed bleached wool proteins, Photochem. Photobiol., 82(2), 551–557 (2006). (6) L. J. Kirschenbaum, X. Qu, and E. T. Borish, Oxygen radicals from photoirradiated human hair: an ESR and fluorescence study, J. Cosmet. Sci., 51(3), 169–182 (2000). (7) K. R. Millington and L. J. Kirschenbaum, Detection of hydroxyl radicals in photoirradiated wool, cotton, nylon and polyester fabrics using a fluorescent probe, Color. Technol., 118(1), 6–14 (2002).
709 UV OXIDATION (8) J. M. Marsh, R. Iveson, M. J. Flagler, M. G. Davis, A. B. Newland, K. D. Greis, Y. Sun, T. Chaudhary, and E. R. Aistrup, Role of copper in photochemical damage to hair, Int. J. Cosmet. Sci., 36(1), 32–38 (2014). (9) C. Cabrera, R. Giménez, and M. C. López, Determination of tea components with antioxidant activity, J. Agric. Food Chem., 51(15), 4427–4435 (2003). (10) W. Koch, J. Zagórska, Z. Marzec, and W. Kukula-Koch, Applications of tea (Camellia sinensis) and its active constituents in cosmetics, Molecules, 24(23), 4277 (2019). (11) A. Kaur, P. Thatai, S. Sharma, and B. Sapra, Green tea extract in microemulsion: Stability, dermal sensitization and efficacy against UV induced damages, Int. J. Pharm. Pharm. Sci., 8(Suppl 1), 1–8 (2016). (12) M. A. Alboreadi, M. M. Al-Najdawi, Q. B. Jarrar, and S. Moshawih, Evaluation of hair growth properties of topical kombucha tea extracts, Adv. Tradit. Med. 22, 151–161 (2022). (13) C. Nualsri, N. Lourith, and M. Kanlayavattanakul, Development and clinical evaluation of green tea hair tonic for greasy scalp treatment, J. Cosmet. Sci., 67(3), 161–166 (2016). (14) J. Huang, C. L. Hastings, G. P. Duffy, H. M. Kelly, J. Raeburn, D. J. Adams, and A. Heise, Supramolecular hydrogels with reverse thermal gelation properties from (oligo)tyrosine containing block copolymers, Biomacromolecules, 14, 200–206 (2013). (15) F. W. Ward, The absorption spectra of some amino acids, Biochem. J., 17, 898–902 (1923). (16) M. A. Rogers, L. Langbein, S. Praetzel-Wunder, H. Winter, and J. Schweizer, Human hair keratin- associated proteins (KAPs), Int. Rev. Cytol., 251, 209–263 (2006). (17) C. R. Robbins and C. H. Kelly, Amino acid composition of human hair, Text. Res. J., 40, 891–896 (1970). (18) D. Creed, The photophysics and photochemistry of the near-UV absorbing amino acids–1. Tryptophan and its simple derivatives, Photochem. Photobiol., 39(4), 537–562 (1984). (19) B. Kierdaszuk, I. Gryczynski, A. Modrak-Wojcik, A. Bzowska, D. Shugar, and J. R. Lakowicz, Fluorescence of tyrosine and tryptophan in proteins using one- and two-photon excitation, Photochem. Photobiol., 61(4), 319–324 (1995). (20) R. V. Benasson, E. J. Land, and T. G. Truscott, Excited States and Free Radicals in Biology and Medicine (Oxford University Press, Oxford, United Kingdom, 1993). (21) J. W. Longworth, “Luminescence of Polypeptides and Proteins,” in Excited States of Proteins and Nucleic Acids, R. F. Steiner and I. Weinryb, Eds. (Plenum Press, New York, 1971), pp. 319–484. (22) C. Giulivi, N. J. Traaseth, and K. J. A. Davies, Tyrosine oxidation products: analysis and biological relevance, Amino Acids, 25(3–4), 227–232 (2003). (23) P. Groves, J. M. Marsh, Y. Sun, T. Chaudhary, and V. Chechik, Effect of humidity on photoinduced radicals in human hair, Free Radicals Med. Biol., 121, 20–25 (2018). (24) P. Groves, J. Huang, A. Heise, J. Marsh, and V. Chechik, Molecular environment and reactivity in gels and colloidal solutions under identical condition, Phys. Chem. Chem. Phys., 22(21), 12267–12272 (2020). (25) D. I. Pattison, A. S. Rahmanto, and M. J. Davies, Photo-oxidation of proteins, Photochem. Photobiol. Sci., 11(1), 38–53 (2012). (26) W. A. Yehye, N. A. Rahman, A. Ariffin, S. B. A. Hamid, A. A. Alhadi, F. A. Kadir, and M. Yaeghoobi, Understanding the chemistry behind the antioxidant activities of butylated hydroxytoluene (BHT): a review, Eur. J. Med. Chem., 101, 295–312 (2015). (27) J. M. Marsh, M. G. Davis, M. J. Flagler, Y. Sun, T. Chaudhary, M. Mamak, D. W. McComb, R. E. A. Williams, K. D. Greis, L. Rubio, and L. Coderch, Advanced hair damage model from ultra-violet radiation in the presence of copper. Int. J. Cosmet. Sci., 37(5), 532–541 (2015). (28) K. R. Millington, Photoyellowing of wool. Part 2: Photoyellowing mechanisms and methods of prevention, Color. Technol.,122(6), 301–316 (2006). (29) E. Fernández, B. Martínez-Teipel, R. Armengol, C. Barba, and L. Coderch, Efficacy of antioxidants in human hair, J. Photochem. Photobiol. B, 117, 146–156 (2012).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)