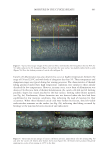
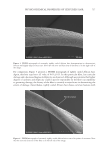
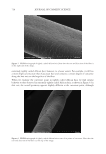
706 JOURNAL OF COSMETIC SCIENCE TEA EXTRACTS In this work, tea (C sinensis) was the chosen botanical antioxidant to test its ability to scavenge reactive oxygen species in the presence of UV radiation. It is rich in polyphenols and has been shown to deliver benefits to both skin and hair (10). Several publications have studied the use of tea extracts as protection against UV-induced skin damage and in the development of sunscreen products (11,30). Tea extracts have also been studied for their impact on hair growth (12) and sebum reduction (13). In an earlier study, 16 tea extracts were chosen, and their antioxidant abilities were tested by ORAC assay (31). This assay is commonly used to measure the antioxidant capacities of foods such as teas, fruits, and vegetables, and it measures inhibition of peroxyl-radical-induced oxidations by the hydrogen-atom-transfer mechanism, a mechanism relevant to human biology and hair. The azo initiator 2,2′-azobis(2-amidinopropane) dihydrochloride is used to generate peroxyl radicals, and a fluorometer is used to measure the rate at which the formed radicals oxidatively degrade an added fluorescent molecule (fluorescein). Table III reports the ORAC data from the 16 tea samples tested and shows that the ORAC scores ranged over three orders of magnitude from 92 to 3,413,500 μM Trolox™ equivalents (TE). Liquid chromatography/mass spectrometry was used to measure the compositions of the 16 tea samples, and a total of 42 compounds were detected, of which 34 were identified full details of the compounds identified are available in Davis et al. (31). The majority of extracts contained low levels of all compounds, with the exception of the four tea samples with BICs 29455, 29458, 29462, and 29465. These four extracts contained similar profiles Table III ORAC Scores for C sinesis (Tea) Extracts* BIC Tea type Form Delivery ORAC score (μM TE/100 g) Standard deviation 29465 Green Powder Made up to 5% 3,413,500 241,123 29458 Green Powder Made up to 5% 1,897,076 45,142 29455 Green Powder Made up to 5% 1,500,536 49,262 29462 Green Powder Made up to 5% 1,454,008 181,366 29459 Green Liquid 10–25% in propylene glycol 108,702 40,091 29471 Green Powder 5% with maltodextrin 71,797 10,341 29464 Green Liquid Unknown conc. in glycerin 46,920 2,969 29463 White Liquid 10% in propylene glycol 42,635 7,840 29469 Green Liquid 5% in glycerin 40,580 23,079 29457 Green Liquid 5% in glycerin 22,861 6,554 29467 White Liquid Unknown conc. in propylene glycol 15,098 2,303 29470 White Liquid Unknown conc. in glycerin 7,980 654 29460 Green Liquid Unknown conc. in glycerin 3,028 62 29456 White Liquid Unknown conc. in glycerin 1,747 174 29466 White Liquid Unknown conc. in glycerin 1,025 186 29461 White Liquid Unknown conc. in glycerin 92 13 *Reproduced with permission from Scientific Research Publishing, Inc. (31).
707 UV OXIDATION of catechin compounds and caffeine (Figure 4), whereas extracts with low ORAC scores contained lower levels of these compounds. A correlation between the ORAC scores and the identified components showed that (−)-epicatechin and procyanidin B had the largest correlation coeffients, and most catechins in the tea samples significantly correlated (p 0.001) with the ORAC scores (31). HAIR UV BIOMARKER EXPERIMENTS Previously published work has shown that the exposure of hair to UV radiation produces protein damage (2) and that MALDI time-of-flight (TOF) mass spectrometry (MS) can be used to identify specific fragments that correlate with total protein damage and UV Figure 4. Protection from damage as measured by UV biomarker intensity (m/z 1,278). *, p 0.05 versus UV-only, Student’s t-test. Reproduced with permission from Scientific Research Publishing, Inc. (31). 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Time (min) 0 50000 100000 150000 200000 250000 300000 1 2 3 4 5 6 7 8 9 10 Figure 4. Chromatogram of tea sample BIC 29465. Major compounds: 1,gallocatechin 2,(−)-epigallocatechin 3, (+)-catechin 4, caffeine (procyanidin B peak lies behind this peak) 5, epigallocatechin gallate 6, (−)-epicatechin 7, gallocatechin gallate 8, (−)-epigallocatechin-3-(3″-O-methyl) gallate 9, (−)-epicatechin gallate 10, (−)-catechin gallate.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)