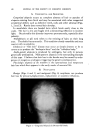
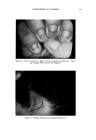
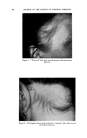
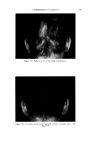
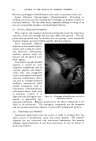
468 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS also know that the area between the curve and vertex "A" has higher vis- cosity, and that the area between the curve and side B-C represents lower viscosities, we gain considerable insight as to how viscosity will be affected by varying all three ingredients in all possible combinations. We can also mark off the line in the triangle (Fig. 6) as was done in Fig. 5 to demonstrate a fourth variable. A more detailed picture of viscosity.variation may be developed if we wish to include the fourth variable, perhaps a fourth ingredient. We merely add a series of curves of constant viscosity (Fig. 7). 60% $o•p.-0il 07o Alcohol /,OYo Water O Clear llguid •2 Cloudy I,c}u,d © Opegue 9el ß Op•oue I,gui& ß C•oua• gel O Clear gel 5oap-0il Physical .ebontie relative fo to % 5o•p-0il Alcohol o 7 Alcohol Water Soap- OiJ• Alcohol •- •h/afer 90• Water Fig. 9A In this graph, each curve represents a different constant viscosity due to the addition of varying amounts of a fourth ingredient. It is easy to pick a particular value of ingredient to give the specific viscosity desired for the final product. This viscosity can be achieved by combining that value of the ingredient with any of the combinations of`4, B and C appearing on the constant viscosity curve. The final choice of the most desirable ratio of .4, B and C will now depend on other factors such as cost, final "feel," texture, stability, etc.
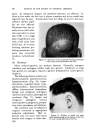
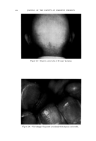
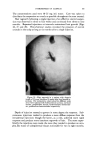
USE OF TERNARY DIAGRAMS 469 ADVANTAGES Or THE SYSTEM The major advantage of combining ternary diagrams with the concept of crossed and paralleled series is to reduce the number of samples required to obtain desired information. Let us illustrate the use of this concept with ternary diagrams to solve a typical problem: finding the most stable W/O emulsion using a particular emulsifier. Such a system may be represented by a ternary diagram as in Fig. 8. Ten samples along a line from "B" to the vertex representing "100 per cent 60% 50•.P 0% •0• Cleo, t' Liclu d C l o-dy Cle• Hazy Opaque 10% ..,qoa. p- Oil 10% 50• Alcohol C larit 7 at 25 ø C Oro nao'hol •-0 % Water c)0%W•ter Fig. 10 water" were first prepared and tested. The most stable one was found at point "C." Using the principle of crossed series, a line from "100 per cent Emulsifier" through point "C" to point "E" was drawn, and a series of ten samples again prepared, more closely spaced, however, and all in the region of points "C" and "E" because the least possible emulsifier was desired. An improvement over "C" was found at point "D." Another improvement was found at point "F" on a line drawn from the "oil vertex" through point "D." The area was now small enough for the use of a small group of paralleled series samples closely spaced around "F." Of these, several for-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)