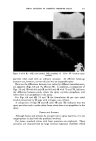
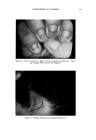
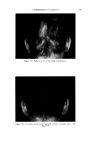
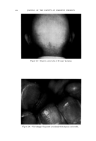
47O JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 60 ø/,, ,5 oap-Oil 0% Alcoho• XkO% Vdaq'e r Cl•ar Ljquid Clear HQzy 10% O,l-$o.p Clarity at •0% Alcohol O%Alcohol 40% Water 90% Water Fig. 10A mulas (quite closely spaced) were found to be best for stability. Fur- thermore, they were definitely the best possible for the system, not just the most satisfactory produced so far. The problem just demonstrated has been oversimplified to illustrate more clearly the procedure of employing the ternary diagram as a tool in formulation work. In practice, more than three variables will be en- countered. Therefore, to demonstrate the value of the method in greater detail we have applied it to a practical, but much more complex problem. A C•,EAR GE•, A solid clear gel with emollient properties was desired one as inexpensive as possible. Emollient products often contain oily materials incompatible with water, yet water is desirable for reasons of economy. The literature showed that certain proportions of soap and alcohol form clear gels with water and oil. Certain alkali metal soaps were noted as being most effi- cient for this purpose. However, these clear gels were quite susceptible to changes in temperature, a factor which had to be considered as the formula evolved.
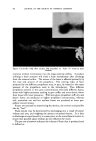
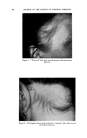
USE OF TERNARY DIAGRAMS 60% $o•p- 0il 0% Alcohol /•OYo Woter 471 loud 3 , Cle Clcer L quicl I0% Soap-Oil Clarity a•' /t.5øC •o% Soap-oH .50% Alcohol 0% Alcohol •0% WaTer •0% Water Fig. 10B The variables forming the basis of our ternary diagram were water, al- cohol and a predetermined mixture of soap and oils having desirable cos- metic properties. On the diagram, the sum of these variables always totals 1• per cent. Since the amounts of antiseptics, color, perfume, etc., are usually predetermined, they were kept constant in all formulations tried. This constant quantity should be added to the sum of the variables above in order to calculate a final 100 per cent formula. The diagram itself was prepared as shown in Fig 9. A series of eight samples along a line from point "A" to the vertex representing 100 per cent water was prepared, resulting in a reasonably good sample at point •7. Samples on either side of this point were more cloudy, and changed to c/ear liquids as they approach point "A." This suggested that a cloudy band would appear on the chart between the desired clear gel and the clear liquid area. Using the principle of crossed series, nine samples were prepared along a line through •7 to the "Soap-Oil" vertex. Three possible products were found at B2, Ba and B4. One of them was brilliantly clear and seemed quite suitable on initial inspection. However, more samples were necessary for general stability evaluation. Using the principle of paralleled series, four
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)