730 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS due to the heat treatment of"tempering." According to Andersson, "it is a question of directing crystallization through a rational process of tempering and cooling in order to get the fat phase to solidify inside the chocolate paste in the stable crystalline •-form (7). This treatment is rather lengthy and is performed on chocolate poured in a pasty state into molds placed on vibrators. The method seems difficult to apply to suppositories in pharmaceutical laboratories, because fatty substances are poured in a fluid state and tempering chambers are not available. Detailed examination of the different forms of "bloom" makes it possible to demonstrate the different reasons for these phenomena and perhaps sug- gest an effective remedy. It is, however, useful to review first some basic concepts about the crystallography of the fatty substance and the physical state of solid fatty substances and to specify what is meant by "change of volume." Crysta#ography of Fatty Substances Triglycerides exist in different polymorphous forms. For the sake of simplicity, it is reasonable to agree with Lutton (8)--to the exclusion of any other theory--that there are three polymorphous forms, designated by the symbols, a, •' and •, listing them in the order of rising melting points, rising stabilities and rising densities. When a triglyceride is melted at temperatures above 70øC and then suddenly cooled, a so-called "tempered" fatty substance is obtained, the crystals of which are in the a-form. The latter can change rather rapidly to give the •'-form and then far more slowly to yield the •-form. It is then said that the fatty substance is "tempered." It should be noted in particular that, at a given temperature, the most stable form has a higher density than the others. Physical State of Solid Fatty Substances Although it is generally considered that a suppository mass passes di- rectly from the solid state to the molten state, this is not entirely true. Cocoa butter can be considered entirely liquid at 40øC. It is then neces- sary to cool it to a low temperature (generally below 0øC) to be certain that it is composed entirely of solid crystals. Between these limits, e.g., at 25øC, cocoa butter should be regarded as a "plastic solid," i.e., it con- sists of crystals impregnated with a liquid phase. The solid crystals are not separated from each other but constitute a skeleton like felt. The inter- stitial liquid also serves as solvent of part of some of the solid components. The relative proportions of solid crystals and liquid phase at different temperatures can be studied directly by dilatometry or evaluated indi- rectly by measurement of consistency, e.g., by means of micropenetrom- etry with a falling needle (9, 10).
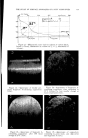
THE STUDY OF SURFACE ANOMALIES OF FATTY SUBSTANCES 731 /•o• Figure 4.--Dilatometre (Dilatom- &re = Dilatometer Roda = Ground Montage = Set-up Pince = Clamp Vide = Vacuum Bois = Wood all dimensions in mm.). In the specific case under discussion, a crystalline skeleton might change in volume as a result of polymorphic modifications at constant tempera- tures, while the impregnating liquid apparently could remain at constant volume it can then be anticipated that certain strains and migrations will occur within the plastic solid. This concept suggested a study of the variations in volume of the solid fatty substance. Specifically, it seemed important to determine the time required for completion of the variations in volume, i.e., the polymorphic transformations. For this purpose, dilatometry at constant temperature was developed which makes it possible to analyze variations in volume due to polymorphism, to the exclusion of any phenomenon due to temperature variations. Surface defects appear readily at constant temperature. Temperature variations are not at all necessary, as has been claimed by certain authors. EX PERIMENTA L Dilatometry at Constan/ Temperature The procedure described here differs from the standard volumetric proc- esses of dilatometry because the latter always seek to reveal variations
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)