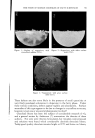
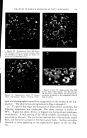
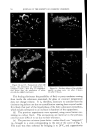
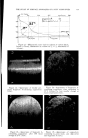
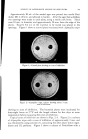
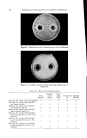
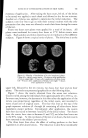
768 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS microbial agent which restilts in a significant reduction of the microbial flora, a decrease in the amount of scalp desquamation occurs. b vidence fi r this decrease was obtained when samples of human dandruff were removed using a hand vacuum cleaner and weighed prior to, during and after treat- ment of the scalp. The average weights of scurf samples from nine subjects decreased Gllowing the application of an antimicrobial agent. A relationship exists between the presence of a scalp microbial flora and the extent of epidermal desquamation. The complete absence of a micro- bial flora does not result in an absolute cessation of scaling. The reduction varies for different individuals between 9 and 55%. It was possible to eliminate the microbial flora after one or two days of treatment. The result of this change was not manifested in measurable scurf reduction for two weeks. This lag indicated that the scalp epidermal desquamation is a physiological process which slowly adjusts itself to environmental changes. Furthermore, when treatment was stopped, scurf production remained at a low level for a much longer period than was required for the microbial flora to become re-established. It would appear that the most effective antidandruff preparations are those which have a twofold action: that of providing a microbiologically free scalp and that of physically removing adhering dandruff scales. A medicated shampoo has both of these actions. The daily application of a preparation containing an antimicrobial agent to supplement the action of the shampoo is recommended. All the subjects participating in the experi- ment described in this paper remarked upon the beneficial effects of the treatment. The scalp felt clean and was entirely free of irritation and itching as long as it was in a state free of any significant microbial flora. (Received June 26, 1964) REFERENCES (1) Roia, F. C., VanderWyk, R. W., and Beal, J. A., •. 8oc. Cosmetic Chemists, 14, 81 (1963). (2) Beal, J. A., Master of Science Thesis, Massachusetts College of Pharmacy, Boston (1962). (3) Epstein, J. M., J•id. (1963). (4) DiM•nna, M. E., y. Pathol. Bacteriol., 68, 98 (1954). (5) MacKee, G. M., and Lewis, G. M., y. Invest. Dermalol., 1, 131 (1938). (6) MacKee, G. M., Lewis, G. M., Pinkerton, E. M., and Hopper, M. E., J•id., 2, 31 (1939). (7) Pachtman, E. A., Vicher, E. E., and Brunner, M. J., J•id., 22, 389 (1954). (8) Lubowe, I. I., Proc. $ci. Sect. Toilet Goods •lssoc., 28, 18 (1957). (9) Spoor, H. J., I•id., 23, 27 (1955). (10) Gordon, M. A., M_ycolojria, 43, 524 (1951).
BOOK REVIEWS IDENTIFICATION AND ANALYSIS OF SURFACE-ACTIVE AGENTS BY IN- FRARED AND CHEMICAL METHODS, Text and Spectra vols. by Dieter Hummel. Translated by E. M. Wulkow. Interscience Publishers, Inc., New York, N. Y. 1964. Text--386 pages spectra--IS6 pages, illustrated and indexed. Price $12. The determination of the surfac- tant components present in such complex mixtures as cosmetic emul- sions, waxes and polishes, insecti- cide emulsions, petroleum products and detergents is a task of no mean proportion. The first and possibly most critical step is the quantitative separation of the sur- factants from the other components of the system. Here, the analyst must rely on his own resources. He must have sufficient knowledge of the system under consideration to be able to estimate the classes of compounds that are likely to be present, and he must know enough about the physico-chemical prop- erties of these components to be able to devise an effective separa- tion procedure. After he has isolated the surfac- rants, the analyst must decide which of the several hundred dif-- ferent surfactants that are com- mercially available are present in his sample. Some shrewd guesses can be made, depending upon his familiarity with the product, but it is still necessary to separate the surfactant mixture into its com- ponents and to identify them. The standard work dealing with the separation and chemical identi- fication of surfactants is M. J. Rosen and H. A. Goldsmith, Sys- tematic ,4nalysis of Surface ,4ctive ,4gents, Interscience, New York, 1960. If the analyst also wants to include infrared techniques, various collections of spectra, such as that of Sadtler, are available. This raises the question of need for this book, and the author properly replies that much confusion is avoided bymaking the infrared approach as systematic as possible. Further, a combination of infrared and chemical methods saves time and makes the results more reliable. The text volume begins with a brief discussion of surfactants and systems of classification, followed by methods for determining the electrochemical behavior of sur- factants. More than half of the volume is then devoted to the infrared spectroscopy of surfac- rants. This includes sample prep- aration, significance of spectra, and detailed discussions of the spectra of practically every commercially available class of surfactants. The remaining chapters are concerned with chemical methods of analysis, including ion exchange methods. Some 100 pages of tables follow the text, and these will be a consider- able aid to the analyst. They in- clude detailed systematic classifica- tions of surfactants, a list of com- mercial surfactants similar to Mc- Cutcheon but including index num- ber and spectrum reference, and numerous tables dealing with sep- aration processes, chemical tests and physical data. 769
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)