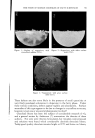
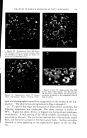
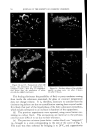
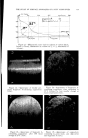
716 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS REFERENCES (1) Daniels, F., Jr., Hopkins, C. E., and Fitzpatrick, T. B., VI.M. VI. virch. Dermatol., 77, 503 (1958). (2) Daniels, F., Jr., in D. B. Dill, Man and Radiant Energy: Solar Radiation, Handbook of Physiology, Sect. 4, Adaptation to the Environment, American Physiological Society, Washington, D.C., 1964, pp. 969-997. (3) Blum, H. F., Eicher, M., and Terus, W. S., vim. y. Physiol., 146, 118 (1946). (4) Blum, H. F., "Sunburn," in A. Hollaender, Radiation Biology II, McGraw-Hill Book Co., Inc., New York, 1955. (5) Hausser, K. W., and Vahle, W., IFiss. ?er6ff. Siernens-Konzern., 6, 101 (13 May 1927). (6) Sanderson, J. A., and Hulbert, E. O., "Sunlight as Source of Radiation," in A. Hollaender, Radiation Biology II, McGraw-Hill Book Co., Inc., New York, 1955. (7) Daniels, F., Jr., virch. Derrnatol., 85, 358 (1962). (8) Daniels, F., Jr., and Imbrie, J. D., y. ]nvest. Derrnatol., 30, 295 (1958). (9) Daniels, F., Jr., Hopkins, C. E., Imbrie, J. D., Bergeron, L., Miller, O., Crowe, F., and Fitzpatrick, T. B., y. ]nvest. Derrnatol., 32, 321 (1959). (10) Daniels, F., Jr., "Quantitative Measurements of the Reactions of Human Skin to Ultx a- violet Light," in B. Chr. Christensen and B. Buchmann, Progress in Photobiology, Proc. of the 3rd lntern. Congr. Photobiology, Elsevier, Amsterdam, 1961. ERRATUM REVERI•CE No. 3 on page 577 of the September 16, 1963, issue should read H. Green, Ind. Eng. Chem. •Inal Ed., 13, 632 (1941).
•. Soc. Cosmetic Chemists, 15, 717-725 (1964) CHEMICAL STRUCTURE AND ANTIMICROBIAL ACTIVITY OF BIS-PHENOLS. IV. BROAD SPEC- TRUM EVALUATION OF 2,2'-METHYLENEBIS (DI CH LOROPH ENO LS) By WILLIAM S. GUMP, PH.D., and GEORGE R. WALTER, PH.D.* ABSTRACT Five symmetrical 2,2'-methylenebis (dichlorophenols) and the asymmetrical 3,4,4',5'-tetrachloro-2,2'-methylenediphenol were in- vestigated for their antimicrobial activity against bacteria, molds and yeasts. The three isomers in which the 6- and 6'-positions adjacent to the hydroxyl groups are not substituted by chlorine were found to exhibit a broader spectrum of activity against the test organisms. The most potent compound was 2,2'-methylenebis (3,5-dichloro. •henot) less active was 2,2'-methylenehis (4,6-dichlorophenol), G-5© (Sindar Corp.), a compound known for many years. It lacked activity against gram-negative bacteria (with the exception of Proteus vulgaris), yeasts and molds. None of the isomers was effective against gram- negative bacteria in presence of soap at a level of 20 •g. mi. A previous study pertained to the bacteriostatic and fungistatic properties of hexachlorophene and its isomers (1). It was reported earlier (1,2) that 2,2'-methylenebis (3,4,5-trichlorophenol) exhibited the greatest activity against gram-positive and gram-negative bacteria. This com- pound is the only isomer of hexachlorophene which has no chlorine sub- stituents adjacent to the hydroxyl groups. It seemed to be of interest to determine whether other bis-phenols with ring hydrogen atoms next to the hydroxyls are more active than those in which these hydrogen atoms are replaced by chlorine atoms. The series of 2,2'-methylenebis (dichloro- phenols) was selected to be suitable for such a comparison. Of the six possible symmetrical isomers, five were synthesized and are listed in Table I with their melting points and chlorine contents. Omitted is 2,2'-methyl- enebis (4,5-dichlorophenol), as no satisfactory method for its preparation * Sindar Corporation, Delawanna, N.J. 717
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)