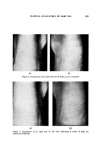
CLINCAL EVALUATION OF BABY OIL 229 and shins) were randomized according to a schedule developed prior to initiation of the study. To be eligible for participation, candidates were re- quired to be free of any dermatologicaI pathology related to systemic diseases and to exhibit severity of symptoms conforming to a rating of 2 or higher on a 5-point rating scale for at least 2 of the 4 specified skin sites as follows. A. Elbows and knees 0=smooth visually and to the touch 1="snow" visible in creases slightly rough to the touch 2--scales and "snow" visible moderately rough to the touch 3=pronounced scaling and roughness to the touch 4=cracking associated with severe roughness to the touch B. Heels 0=smooth visually and to the touch l=rough to the touch with "snow" and cracking just visible 2=rough to the touch cracking and scaling evident 3=quite rough to the touch scaling and cracking pronounced 4=very rough to the touch extreme scaling and cracking C. Tibia 0=smooth visually and to the touch i--smooth to the touch "snow" visible in creases 2=scaling and slight glazing 3=pronounced scaling and slightly rough to the touch 4=severe scaling and cracking very rough to the touch Following the baseline examination, subjects were instructed to utilize at least 2 drops of the baby oil on the designated areas at least twice daily (on arising and at bedtime) during a 4-week study period. While the oil was to be applied sparingly so that it was not recognizable at the times of clinical assessment, they were requested to use enough to cover the test sites com- pletely. Use of other skin conditioning preparations during the study was pro- hibited. The subiects were again rated after 2 and 4 weeks. All examinations were conducted in a uniform manner with elbows and knees bent at a 90 ø angle. Discussions between the investigator and subject were prohibited, and at no time was the investigator made aware of the identity of the treatment side. Finally, photographic documentation of effectiveness was obtained from 20 of the subjects. These individuals had been selected at random and specified by subiect number prior to therapeutic trial. Each of the 4 sites on both the right and left sides of the subiects were photographed at the scheduled exami- nations and again either 24 or 48 hours following treatment withdrawal to allow a determination of duration of baby oil activity.
230 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS RESULTS Valid data were obtained from 106 of the initial 111 participants. Four sub- jeets were excluded from the final statistical analysis because of insuttleient baseline dermatologie involvement and one because of insuttleient rating data due to a missed rating session caused by intercurrent illness. That the initial severity of the dermatologieal signs were comparable for the treatment sites and contralateral untreated sites among the population can be ascertained by review of Table I. In general, skin of elbows, knees, and heels exhibited a greater degree of drying, sealing, and roughness at the out- set of the trials in comparison to shins. Skin moisturizing activities of the baby oil are illustrated in Fig. 1. Severity of the signs was decreased in comparison to baseline readings to a significant extent at all 4 treatment sites. Conversely, untreated sites tended to become more severely afflicted over the 4-week study period. Actually, tibial areas were significantly worse at the 4-week evaluation (P=0.01). Improvement ap- peared to be progressive with continued treatment. However, even after 2 weeks, the degrees of benefit were clearly demonstrable. A summary of the statistical analyses of the data is presented on Table II. Results of evaluations of the 10 individuals who returned to the clinic 24 hours after the baby oil applications were terminated and the additional 10 who reported to the clinic 48 hours later are summarized in Table III. The data suggest that the protectant moisturizing actions of baby oil do not persist for extended periods following termination of therapy, as skin of treat- ed sites tended to return to the pretreatment state during the 24- and 48-hour follow-up periods. Mean scores calculated from raw data and obtained utilizing the described objective methods prove the thesis that the completely nonpolar liquid hydro- Table I Distribution of Sites by Treatment Group and Initial Severity of Dermatologic Signs Site _ Elbow Knee Heel Tibial Area Sever'ty a Treated Untreated Treated Untreated Treated Untreated Treated Untreated 0 5 3 7 9 4 4 6 7 1 24 27 40 ,35 20 20 48 b 49 b 2 53 • 52 • 48 • 50 • 62 • 59 b 43 41 3 20 20 10 .11 18 21 9 9 4 4 4 1 1 2 2 0 0 TOTALS: 106 106 106 106 106 106 106 106 a Median severity. •'S-e protocol for definitions.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)