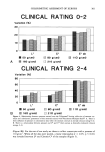
COLORIMETRIC ASSESSMENT OF XEROSIS 305 In previous works we had found that CSSB were satisfactory for most routine work, enjoying a good correlation with clinical evaluations of pathological conditions and bringing unique information concerning the pattern and type of xerosis (1,4). For dry skin, however, a rough correlation that did not reach statistical significance was seen between visual assessment and microscopic scores of CSSB. The lack of significance was due to the fact that microscopic grades 0, 1, and 2 were rated clinically as normal without further distinction. On the other hand, grades 4 and 5 appeared indistinguish- able by visual inspection of the skin alone. Colorimetric measurements of D-Squame © offer an accurate method for quantitative assessment of dry skin. The combination of CSSB and colorimetry of D-Squame © therefore offers the advantages of typing and rating dry skin. These methods are appli- cable to various studies of xerotic skin--for example, to assess the effect of moisturizers, influence of age, etiopathogenesis, etc. REFERENCES (1) C. Pi6rard-Franchimont and G. E. Pi•rard, Les x•roses: Structure de la peau r&che, Int. J. Cosmet. Sci., 6, 47-54 (1984). (2) G. E. Pi•rard, What do you mean by dry skin? Dermatologica, 179, 1-2 (1989). (3) J. Serup, A. Winther, and C. Blichmann, A simple method for the study of scale pattern and effects of a moisturizer. Qualitative and quantitative evaluation by D-squame tape compared with parameters of epidermal hydration, Clin. Exp. Dermatol., 14, 277-282 (1989). (4) C. Pi•rard-Franchimont and G. E. Pi•rard, Assessment of aging and actinic damages by cyanoacrylate skin surface strippings, Am. J. Dermatopathd., 9, 500-509 (1987). (5) M. F. Lukacovic, F. E. Dunlap, S.C. Michaels, M. O. Visscher, and D. D. Watson, Forearm wash test to evaluate the clinical mildness of cleansing products, Jo Soc. Cosmet. Chem., 39, 355-366 (1988). (6) H. Pehamberger, A. Steiner, and R. Wolff, In vivo epiluminescence microscopy of pigmented skin lesions. I. Pattern analysis of pigmented skin lesions, J. Am. Acad. Dermatol., 17, 571-583 (1987). (7) H. P. Soyer, J. Smolle, S. HiSdl, H. Pachernegg, and H. Keri, Surface microscopy. A new approach to the diagnosis of cutaneous pigmented tumors, Am. J. Dermatopathol., 11, 1-10 (1989). (8) J. Serup and T. Agner, Colorimetric quantification of erythema--A comparison of two colorimeters (Lange Micro Color and Minolta Chroma Meter CR~200) with a clinical scoring scheme and laser- doppler flowmetry, Clin. Exp. Dermatol. 15, 267-272 (1990). (9) F. Deleixhe-Mauhin, C. Pi•rard-Franchimont, and G. E. Pi•rard, Introduction au monde de la couleur en m•decine, Rev. Med. Liege, 46, 607-612 (1991).
j. Soc. Cosmet. Chem., 43, 307-312 (November/December 1992) An in vitro method for screening sunscreen formulations for sun protection factor using a full-thickness skin model BARRY T. REECE, DAVID DEEDS, and MICHAEL ROZEN, Biophysical Skin Research Laboratory, Mary Kay Cosmetics, Inc., Dallas, TX 75247. Received April 14, 1992. Synopsis With the aid of a full-thickness skin model, minimum sun protection factors (SPF) of various sunscreens were determined. Sunscreen efficacy was determined by the release of interleukin IL- lot. The data show that this in vitro model is capable of determining minimum SPFs of a formula and may prove useful as a screen in the product development process. INTRODUCTION Sunscreens represent an important category of consumer products, permitting users to enjoy outdoor activities while reducing skin exposure to harmful ultraviolet (UV) light. In the United States, sunscreen products are regulated as over-the-counter drugs, and their safety and sun protection factor (SPF) claims must be established according to the Food and Drug Administration's regulations (1). These regulations require the SPF to be determined under specific conditions using human subjects. Historically, prior to final human SPF testing, in vivo SPF screening studies were performed using guinea pigs. With the suspension or banning of animal testing by many manufacturers of personal care products, a need now exists for reliable in vitro screening assays. Early attempts to define sunscreen protection by in vitro methods involved measuring the transmission of ultraviolet (UV) light through a dilute solution of sunscreen (2) or measuring the UV transmission through a film of sunscreen applied to a quartz glass (3). However, Groves et al. (4) were able to show that these methods cannot be used reliably due to their overestimation of the SPF value. Recently Diffey and Robson (5) described a rapid method for the determination of SPF by measuring the amount of ultraviolet light transmitted through Transpore TM tape with and without sunscreen applied close agreement between the in vitro and in vivo data was observed. Other investigators have measured the transmission of UV light through biological substrates (e.g., skin from hairless mice) as a means of determining SPF values (6,7). Keratinocytes are the principal cells of the skin that respond to UV irradiation, and their involvement in UV-induced inflammatory reactions has been established. Ansel et al. 307
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)