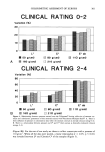
330 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (11) (12) (13) (14) (15) (16) (17) (18) (19) (20) (21) G. E. Nilsson, Measurement of water exchange through skin, Med. Biol. Eng. Comput. 15, 209-218 (1977). J. Pinnagoda, R. A. Tupker, T. Agner, and J. Serup, Guidelines for transepidermal water loss (TEWL) measurement, Contact Dermatitis, 22, 164-178 (1990). H. Tagami, M. Ohi, K. Iwatsuki, Y. Kanamaru, M. Yamada, and B. Ichijo, Evaluation of the skin surface hydration in vivo by electrical measurement, J. Invest. Dermatol., 75, 500-507 (1980). G. E. Nilsson, U. Otto, and J. E. Wahlberg, Assessment of skin irritancy in man by laser Doppler flowmerry, Contact Dermatitis, 8, 401-406 (1982). J. C. Seitz and C. G. Whitmore, Measurement of erythema and tanning responses in human skin using a tri-stimulus colorimeter, Dermatologica, 177, 70-75 (1988). K. P. Wilhelm, C. Surber, and H. I. Maibach, Quantification of sodium lauryl sulfate irritant dermatitis in man: Comparison of four techniques: Skin color reflectance, transepidermal water loss, laser Doppler flow measurement and visual scores, Arch. Dermatol. Res., 281, 293-295 (1989). T. Agner and J. Serup, Sodium lauryl sulphate for irritant patch testing--A dose-response study using bioengineering methods for determination of skin irritation,J. Invest. Dermatol., 95, 543-547 (1990). R. A. Tupker, J. Pinnagoda, P. J. Coenraads, and J. P. Nater, The influence of repeated exposure to surfactants on the human skin as determined by transepidermal water loss and visual scoring, Contact Dermatitis, 20, 108-114 (1989). J. Zhou, R. Mark, T. Stoudemayer, A. Sakr, J. L. Lichtin, and K. L. Gabriel, The value of multiple instrumental and clinical methods, repeated patch applications, and daily evaluations for assessing stratum corneum changes induced by surfactants, J. Soc. Cosmet. Chem., 42, 105-128 (1991). S. Zehnder, R. Mark, S. Manning, A. Sakr, J. L. Lichtin, and K. L. Gabriel, Analysis of irritancy of SLS on human skin: Concentration vs. exposure duration effects, Poster presented at the Annual Scientific Meeting of the Society of Cosmetic Chemists, New York, December 5, 1991. J. A. Faucher and E. D. Goddard, Interaction of keratinous substrates with sodium lauryl sulfate: I. Sorption, J. Soc. Cosmet. Chem., 29, 323-337 (1978).
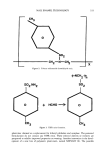
j. Soc. Cosmet. Chem., 43, 331-337 (November/December 1992) Advances in nail enamel technology MITCHELL L. SCHLOSSMAN and ERIC WIMMER, Tevco, Inc., South Plainfield, NJ 07080 (M.L.S.), and SNPE, Bergerac, France (E.W.). Received May 15, 1992. Synopsis Nail enamel is a very esoteric area of cosmetic formulating, related for the most part to paint and lacquer technology. Non-settling or suspension nail enamel was and is a development in cosmetic formulation that represents an excellent example of the application of rheological principles. Suspension nail enamel has undergone many changes from the formulations of the last 30 years. This paper will review some of the past formulations, manufacturing procedures, and evaluation methods, and attempt to discuss the technology of the 1990s. Packaging innovations and advances in nail care lacquer areas such as base and top coat, hardness and strengtheners will be outlined. The presentation will address environmental and governmental issues and the impact they may have on future nail enamel formulations, namely toluene-free, formaldehyde-free, and water-based systems. Nail enamel is a very esoteric area of cosmetic formulating, related for the most part to paint and lacquer technology. There is a remarkable amount of technology packed into that little bottle. Nail enamel is only nominally related to the cosmetic industry in meeting that industry's unique requirements as to governmental regulations and the strange requirement of having a paint product look nice in a tiny glass bottle. Nail enamel formulations are virtually anhydrous, and inherently bactericidal. The manu- facturing process is capital intensive, and involves handling large quantities of hazardous raw materials that are unfamiliar to most cosmetic operations. Nail enamel plants must meet the strict electrical codes for explosion-proof wiring, and the buildings must be designed for explosion venting. For these reasons, cosmetic companies generally do not manufacture their own nail enamel. The vast majority of nail enamel is based on nitrocellulose the structural unit for cellulose is shown in Figure 1. The most important attributes of a nitrocellulose lacquer are that it dries quickly and has a high gloss. Various materials have been suggested as alternative film formers, but none have so far matched nitrocellulose in terms of hard- ness, toughness, and resistance to abrasion. Furthermore, nitrocellulose does not oxidize and polymerize, and it produces films that are waterproof and stable to atmospheric conditions. Finally, nitrocellulose is an extremely good polymer for the wetting of pigments. This is important in effecting good color dispersion. A small pigment load is able to produce clean, bright color with good opacity. Several primary film formers for nail enamel have been tried, namely polyurethanes, polyamides, polyesters, and cel- 331
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)