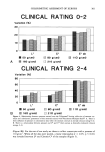
328 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS effects were inversely related to the molar percentage of SLEC-5 or -13 within the various mixtures, and were generally more pronounced in the panelists treated with SLS and SLEC-5. In the group of panelists treated with mixtures of SLS and SLEC-5, half of the correlation coefficients between evaluation methods were significant, whereas they were all significant in the panelists treated with SLS and SLEC-13 (Table VI). COMPARISON WITH STUDIES FROM THE LITERATURE The ability of ethoxylated anionic surfactants to modify the irritancy potential of SLS has been investigated by Rhein and co-workers (5,7) using ether sulfates with varying degrees of ethoxylation. However, no comparable report was found involving ether carboxylates. The series of (C •2--C 14) alkyl ether carboxylates to which SLEC-5 and -13 belong can be compared in terms of its mollifying effect on SLS to that of the (C z2--C z4) alkyl ether sulfates studied by Rhein et alo (5,7). Surfactants from both series are anionics, ethoxylated, and possess on average the same carbon chain length they differ only by the nature of their polar head group: a carboxylate vs a sulfate. In the present study, the intensity of the irritation response was inversely related to the molar percentage of ether carboxylate within the different mixtures of 1.0% SLS:SLEC-5 or -13 even though the total surfactant concentration was increased by the added cosurfactant. Also, SLEC- 13, the higher ethoxylated surfactant, was more effective in modifying SLS irritancy than SLEC-5. Rhein and co-workers (7) performed a 21-day cumulative irritation patch test with various molar ratios of SLS (at 0.25 and 1.0%) and a (C12--C14) alkyl ether sulfate containing 7 moles of ethylene oxide (AEOS-7EO). They demonstrated that addition of AEOS-7EO to a constant concentration of SLS resulted in a significant reduction in erythema. Rhein et al. (5) also reported in vitro findings using a stratum corneum denaturation model measuring membrane swelling induced by mixtures of 1.0% SLS and 1.0% of a (C12--C14) alkyl ether sulfate containing 3 or 6 moles of ethylene oxide (AEOS-3EO or -6EO). Mixtures of the ether sulfates with SLS caused significantly less swelling than 1.0% SLS alone. AEOS-6EO (i.e., the higher ethoxylated surfactant) was more effective than AEOS-3EO at inhibiting swelling. MECHANISMS FOR REDUCTION OF SLS IRRITATION POTENTIAL The reduced skin response observed with mixtures of SLS and milder surfactants may be explained by several possible mechanisms (1,2). Most likely, formation of mixed mi- celies occurs with the addition of a milder cosurfactant (i.e., SLEC-5 or -13), resulting in a reduction in the critical micelle concentration (CMC) of SLS. This brings about a lowering of the irritating SLS monomer concentration, hence producing a milder system (3,4,7,21). It has been proposed and demonstrated that the surfactant monomers rather than the micelles interact with the skin to elicit irritation (3,4,7,21). Micelies are unlikely candidates to penetrate the keratin because of the combination of size and energy limitations (7). The greater effectiveness of SLEC-13 in relationship to SLEC-5 at reducing SLS irritation potential may result from several phenomena. In anionic surfactants, ethoxylation in- creases the hydrophilicity that counterbalances the hydrophobic effect of the alkyl
ASSESSING REDUCTION OF SLS IRRITATION 329 portion (8). A higher degree of ethoxylation also results in a higher reduction in electrostatic repulsion (6). Therefore, it is believed that SLEC-13 produced a greater reduction in the CMC of SLS and a greater lowering in the irritating SLS monomer concentration than did SLEC-5. CONCLUSIONS An in vivo method involving a two-hour occlusive patching procedure on five consecutive days together with multiple instrumental measurements was validated for its ability to assess reduction of SLS irritancy by added mild surfactants. By using this approach, both SLEC-5 and -13 exerted a mollifying effect on SLS, but SLEC-13 (the higher ethoxylated surfactant) was clearly demonstrated to be more effective in reducing the irritancy potential of SLS than SLEC-5. Because of the specificity and sensitivity of the method, valuable data for product formulation can be obtained regarding 1) the relative effec- tiveness of various added surfactants in reducing SLS irritancy, and 2) the SLS:cosur- factant proportions needed to achieve specified levels of irritancy reduction. ACKNOWLEDGMENTS The authors would like to thank Hyman Menduke, Ph.D., of Thomas Jefferson Uni- versity, Philadelphia, Pennsylvania, for his help on the data system and the statistical analyses. Appreciation is extended to Dominique Ribola, Ph.D., and Joan Day of Hoechst Celanese Corp., Charlotte, North Carolina, for supplying the test materials. The authors are also grateful to Richard Hershman, Ph.D., of Biosearch, Inc., Phila- delphia, Pennsylvania, for his comments and discussions during the course of this research. REFERENCES (8) (9) (10) (1) R. L. Goldemberg and L. Safrin, Reduction of topical irritation, J. Soc. Cosmet. Chem., 28, 667-679 (1977). (2) R. L. Goldemberg, Anti-irritants,J. Sac. Cosmet. Chem., 30, 415-427 (1979). (3) J. Garcia Dominguez, F. Balaguer, J. L. Parra, and C. M. Pelejero, The inhibitory effect of some amphoteric surfactants on the irritation potential of alkylsulphates, Int. J. Cosmet. Sci., 3, 57-68 (1981). (4) K. Miyazawa, M. Ogawa, and T. Mitsui, The physico-chemical properties and protein denaturation potential of surfactant mixtures, Int. J. Cosmet. Sci. 6, 33-46 (1984). (5) L. D. Rhein, C. R. Robbins, K. Fernee, and R. Cantore, Surfactant structure effects on swelling of isolated human stratum corneum, J. Sac. Cosmet. Chem., 37, 125-139 (1986). (6) J. C. Blake-Haskins, D. Scala, L. D. Rhein, and C. R. Robbins, Predicting surfactant irritation from the swelling response of a collagen film, J. Sac. Cosmet. Chem., 37, 199-210 (1986). (7) L. D. Rhein, F. A. Simion, R. L. Hill, R. H. Cagan, J. Mattai, and H. I. Maibach, Human cutaneous response to a mixed surfactant system: Role of solution phenomena in controlling surfactant irritation. Dermatalagica, 180, 18-23 (1990). U. K. Charaf and G. L. Hart, Phospholipid liposomes/surfactant interactions as predictors of skin irritation. J. Sac. Cosmet. Chem. 42, 71-85 (1991). J. Day, Application Chemist, Hoechst Celanese Corp., Charlotte, NC, personal communication (1991). P. J. Frosch and A.M. Kligman, The soap chamber test,J. Am. Acad. Dermatal., 1, 35-41 (1979).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)