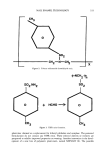
332 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS H H OH CH 2 CH OH H H 0 H H OH H CH20H H OH Figure 1. Structural unit of cellulose. lulosics such as cellulose acetate isobutyrate (CAB), cellulose acetate, cellulose acetate propionate (CAP), and ethyl cellulose (EC). Generally the aforementioned polymers exhibited poor adhesion to the nails or soft films. The advantages of these replacements are low flammability, no discoloration, and solubility in low-cost solvents'. Another development was a nitrocellulose-free nail lacquer (1) comprising copolymers of hydro- phobic and hydrophilic monomers such as isobornyl methacrylate and diacetone acry- lamide, respectively, and applied to nails. A waterproof film having a high gloss was provided. A more recent patent discloses a nail lacquer free from water and nitrocellu- lose, containing alkyl(meth)acrylate and hydroxyalkyl(meth)acrylate in solution (2). A nail lacquer having good gloss and adhesion is claimed. Secondary resins are added to nitrocellulose lacquers to improve application, wear, and gloss properties. The most widely used are the toluene sulfonamide formaldehyde (TSFR) type, as shown in Figures 2 and 3. TSFR is a low-molecular-weight material, used as a filling resin in nail lacquers for improving the quality of the film (adhesion, gloss, flexibility...). There has been some controversy about the use of TSFR because there is a small amount of residual formaldehyde. TSFR has been identified as a potential sensitizer for a small number of nail lacquer users. Polyesters (3), nylon, acrylic ester oligomers, alkyds, etc., have been used as replacements (4). None of the aforementioned have replaced TSFR resin as a secondary resin for nitrocellulose lacquers. In an attempt to identify in the TSFR molecule what kind of function is responsible for its attributes, a study was conducted (5), the purpose of which was to help design a new improved resin for nail enamel. In summary, the analysis of TSFR showed clearly that its commonly accepted representation of a polymeric structure is at best an oversimplification of what is a mixture of oligomers having functional end groups. From that study, the synthesis of a new family of resins, the aryl alkyl sulfonyl urethanes (6), were developed. A structural formula is shown in Figure 4. Another secondary resin derived from para toluene sulfonamide is toluene sulfonamide epoxy resin (7). Plasticizers are used to make the polymer film compatible with the nail. They make the film more flexible. Dibutyl phthalate and camphor are the most widely used plasticizers for nitrocellulose films. There has been a question as to whether or not phthalates act as sensitizers for some nail enamel wearers. A recent patent discusses nail enamels con- taining glycerol, glycol, or citrate esters (9). Glycerol tribenzoate is an example of a
NAIL ENAMEL TECHNOLOGY 333 o ß CH2--N--CH 2 ,, Figure 2. Toluene sulfonamide formaldehyde resin. x •-NCH2 '•n i SO 2 NH 2 SO 2 0 -!'- HCHO • 0 CH 3 CH 3 Figure 3. TSFR resin formation. plasticizer claimed as a replacement for dibutyl phthalate and camphor. The patented formulations do not contain any TSFR resin. These selected diesters or triesters are purported to exhibit improved properties on wearing. Another innovation is the devel- opment of a new line of polymeric plasticizers, named NEPLAST (9). The possible
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)