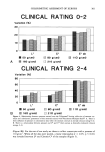
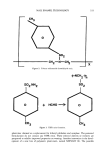
336 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS area. Nail lacquers containing Kevlar ©, gelatin, acrylates, cyano acrylates, microcrys- talline cellulose, and other aldehydes in place of formaldehyde have entered the mar- ketplace. The cosmetics industry faces some tough challenges in the 1990s. Every phase of the industry--from product creation through distribution--is being scrutinized. As environmental consciousness rises, there will be more pressure put on the industry to achieve environmentally friendly products. Although containing solvents, a recent study discussed the development of a w/o emulsion-type nail enamel to avoid dehydration from the nail by supplementing water to the nail (20). Finally, a water-dilutable nail polish was developed containing a mixture of polyurethanes and/or vinyl and/or acrylic esters (21,22). These nail enamels for the most part are of the peelable type. Water-based nail polish may no longer be the "impossible" dream. By the turn of the century, I foresee that the nail polish chemist will devise a water-based nail enamel system that has good adhesion, gloss, and drying qualities. A large number of the colorants used in solvent- based nail lacquers are also usable in water-based nail polish. However, care should be taken to see that the colorant is compatible with water. Organic colorants and pigments, inorganic pigments, and pearlescent materials may be used. As in solvent-based nail lacquers, colorant in the form of a dispersion must be utilized. Unlike in solvent-based nail lacquers, dispersing agents and wetting agents are often used as surfactants to help disperse uniformly the hydrophobic organic pigments. Inorganic pigments are naturally hydrophobic and are easily dispersed in an aqueous system. To achieve a product based on water and equivalent to a conventional solvent-based nail enamel, we will more than likely require a new range of polymers and coalescing and cross-linking agents. Envi- ronmentally safe nail enamel continues to be a very real challenge for the cosmetic chemist in the 1990s. REFERENCES (1) E. S. Abrutyn (to Wicken Products, Inc.), Europ. Pat. Appl. O. 085,370 (1982). (2) B. Langla, J. Mondet, and C. Papanttmiou (to L'Oreal), U.S. Patent 5,057,312 (1991). (3) M. L. Schlossman (to Tevco, Inc.), U.S. Patent 4,301,046 (1981). (4) M. L. Schlossman, Modern nail enamel technology, J. Soc. Cosmet. Chem., 31, 29-36 (1980). (5) V. Chiva, M. Lecacheur, R. Faure, R. Gallo, and E. Wimmer, Structure of toluene sulfonamide formol resins. Molecular models from mechanistic schemes and analytical results, J. Appl. Polym. Sci. (submitted). (6) M. Lecacheur, J. Mutterer, and E. Wimmer, Fr. Patent 9202486 (1992). (7) L. X. Mallavarapu, U.S. Patent 4,996,284 (1991). (8) A. Castrogiavanni, R. W. Sandewicz, and S. W. Amaro (to Revlon, Inc.), U.S. Patent 5,066,484 (1991). (9) Personal communication, SNPE, Dept. Cosmetiques, Mar. 1992. (10) F. W. Busch, Jr. (to Chesebrough-Pond's Inc.), U.S. Patent 3,864,294 (1975). (11) A.M. Kuritzkes (to Mearl Corp.), U.S. Patent 3,422,185 (1969). (12) F. W. Busch, Jr. and M. G. Brookins, Thixotropic flow in cosmetics: Utility and evaluation, SCC New England Chapter Meeting, Boston, April 25, 1974. (13) R. L. Socci, A. A. Ismailer, and A. Castrogiavanni (to Revlon, Inc.), U.S. Patent 4,832,944 (1989). (14) Personal communication, Kobo Products, Inc., Mar. 1992. (15) R. A. Weber, C. Christopher, and J. A. Penicnak (to L'Oreal S.A.), Eur. Patent Appl. EP 479,669 (1992). (16) T. Ikeda, T. Kobuyashi, C. Tanaka, Y. Fujiyama, T. Ozawa, and T. Mitsui, Development of highly safe nail enamel, J. Soc. Cosmet. Chem. Japan, 22, 25-34 (1988).
NAIL ENAMEL TECHNOLOGY 337 (17) J. M. Boisserie, F. La Forest, C. Sanderson, F. Scheuber, and E. Wimmer, Fr. Patent 900859 (1989). (18) E. Wimmer and M. Schlossman, The effect of toluene on the properties of suspension nail polish, 16th IFSCC Congress Preprints, 2, 18-30 (1990). (19) D. Papas and H. J. Larsen (to Amalia Inc.), U.S. Patent 5,093,108 (1992). (20) L. Dell'Aquila, U.S. Patent 5,045,309 (1991). (21) D. So. So (to Hwo Young Park), U.S. Patent 4,903,840 (1990). (22) K. Yamazaki and M. Tanaka, Development of a new w/o emulsion-type nail enamel, 16th IFSCC Congress Preprints, 1, 464-495 (1990). (23) R. Janda, Ger. Patent Appl. P3931237.2 (1990). (24) D. Koch and R. Rassek (to Cosmolab, Inc.), U.S. Patent 5,120,529 (1992).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)