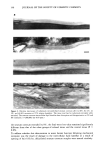
156 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS sample. Figure 1 is the schematic diagram of the ATR technique. ATR-infrared spec- troscopy is a surface analysis technique. The interaction of the light with the sample, and therefore the intensity of the spectral bands, depends on the depth of penetration, which in turn is determined by the following equation: dp = 2-rr (sin20 2 ,]/2 -- n21 ) dp = depth of penetration K• = K/n• • = the wavelength of the radiation n• = the refractive index of the prism (for ZnSe, n• is 2.4) 0 = the angle of incidence n l = the refractive index of the sample (for skin, n l is approximately 1.5) and nl• = nl/n •. Most of the experiments for this study used a ZnSe prism with a 45 ø angle of incidence. These parameters dictate a penetration depth of about 1.0 •m for energy of 6 •m wavelength. N-PERDEUTEROLAUROYL SARCOSINE AS THE MODEL Initial efforts to detect substantivity on skin employed non-deuterated sodium lauroyl sarcosinate. The infrared spectra of human skin and (H23) lauroyl sarcosine are presented in Figure 2. The spectra of skin and the surfactant are quite similar. The surfactant spectrum does not exhibit any unique band that is not overlapped by the bands in the skin spectrum, which renders the direct identification of the surfactant on the skin surface difficult. Computer-aided subtraction of infrared spectra of materials that are similar to each other has been documented in literature as a procedure to identify the minor differences between them. Such efforts may prove successful for establishing the sorption of (H23) lauroyl sarcosinate on skin and is worth pursuing in the future. However, at the time when we had begun this research, we needed direct evidence for the presence of the surfactant on skin. Deuteration of the surfactant offered a way of SOURCE SAMPLE [DETECTOI Transmission IR Spectroscopy SOURCE ystal DETECTOR SAMPLE Depth of Penetration Attenuated Total Reflectance IR Spectroscopy Figure 1. IR sampling techniques. In ATR spectroscopy, IR energy reflected by the crystal interacts with the sample placed in contact with the crystal.
N-LAUROYL SARCOSINATE 157 120.0 80.0 %T 40.0 0.0 I I I • I • I 4000 3000 2000 1400 750 Figure 2. Infrared transmittance spectra of human skin (1) and lauroyl sarcosine (2). altering the surfactant so that it could be detected on the skin surface. It is assumed that isotopic substitution (replacing hydrogen atoms with deuterium atoms) of the surfactant does not significantly change its surface-active properties. Substitution of the hydrogen atom in a H-X bond by a deuterium atom (D, the hydrogen isotope with mass number 2) shifts the H-X vibrational frequency (6). For a simple diatomic molecule, e.g., HC1, the frequency of vibration of the H-C1 bond is related to the bind strength and the masses of the atoms involved by the following expression: tOe = 1307 cm -• toe -- the frequency in cm- • f' = the force constant and p,' = the reduced mass = m•m2/(m • + m2). (m•, m2 = masses of the atoms involved in the vibration.) If the bond strength (f') is assumed to remain unchanged with the isotopic substitution, the ratio of the frequencies of the band position before and after the substitution is given by the equation: toHC1 .•/I,i, DC1 to--•-•- •/ixHC 1 • V • Based on the above equations, a frequency shift by a factor V'1.86 is calculated for the substitution of a hydrogen atom in a C - H bond by a deuterium atom. Lauroyl sarcosine exhibits the C - H stretches in the 3000 cm- •-2800 cm- • region. It was anticipated, --1 therefore, that deuterolauroyl sarcosine would exhibit bands in the 2050-2200 cm range where the spectroscopic interference due to skin is low. Figure 3 is the IR spectrum of N-perdeuterolauroyl sarcosine sodium salt. The bands at 2090 cm- • and
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)