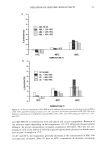
SURFACTANTS AND BLOOD CLEANSING BY SLS SOLUTIONS 103 blood cells to lyse. This method was used to determine the amount of hemoglobin released into the solution by the lysed blood cells. The percent transmittance (%T) at 550 nanometers was determined for each of the hemoglobin solutions using a Perkin- Elmer Lambda 3 UV/VIS spectrophotometer with a tungsten light source. Standard curves were prepared for each surfactant concentration, as well as for each new blood sample. These standard curves were based on blood dilutions and their respective percent transmittances. All of the standard curves were essentially linear, with correlation coefficients close to 1. By using standard curve values, the percent transmittance was converted to milligrams of hemoglobin per milliliter of solution. The percentage of blood removed was calculated by dividing the mg/ml of hemoglobin by 6.25, which was the theoretical amount of hemoglobin delivered in the 50-pl aliquot of blood, and multiplying by 100. All repetitions in an individual series were averaged, with the averages for each sample based on at least ten trials. DISCUSSION Figures 1 through 9 plot the % blood removal vs the concentration of surfactant in the sodium chloride solutions. These curves provide information that can be used to explain the chemical interaction between the sodium chloride in solution and the surfactants. SODIUM LAURYL SULFATE The pH of the 0.9% NaCI/SLS and 2.0% NaCI/SLS solutions (Figures 1, 2) remained between 6.0 and 7.0. The douche solutions (Figures 3, 6, and 9) maintained a pH of 3.32 to 3.38 due to the acetic acid (vinegar) in solution. In acidic solutions, the SLS is unable to ionize to a great degree, restricting its surface-active capabilities. The surface-active anions of SLS, which are hydrophobic alkyl chains with a negative ion attached to one end, form micelles that attract the polar water molecules less than the small, highly polar Na + and C1-. Blood cells are affected by the water around them and, depending on the nature of the solution, the cells can lyse or crenate. The SLS micelies interact and compete with the blood cell membrane and prevent the water from main- taining the pressure outside of the blood cell. Theoretically, the non-polar portions of the surfactant molecules are attracted to the cell membrane by van der Waals forces, with the phospholipid portion entering the cell membrane. This would decrease the cell's permeability to water. As the water pressure outside the blood cell decreases, the pressure inside the cell causes the blood cell to lyse and release its contents into solution. Once in solution, the biological contents of the cell are denatured, and SLS micelles are then capable of removing this denatured material from the cloth. The cleansing drops off as the SLS concentration increases to 0.08% (Figure 1), and the cleansing then increases for the next two successive SLS concentrations. SLS alone in aleionized water is able to cleanse the denatured cellular material from the cloth, but at the lower SLS concentrations no lysing occurs. At these lower concentrations of SLS, the sodium chloride is responsible for the blood removal. As the concentration of SLS increases, more surface-active artions enter the solution. This
104 JOURNAL OF COSMETIC SCIENCE lOO 90 80 o 70 E o 60 o 50 40 30 0.00 I I I I 0.02 0.04 0.06 0.08 % SLS ß NaC1 Solution/SLS O DI Water ß DI Water/SLS Figure 1. 0.9% NaC1/SLS solutions. I 0.10 0.12 increases the amount of micelies within the solution, thereby reducing the solution's ability to remove the blood. At higher concentrations of SLS, the blood cells are being lysed by the SLS, and sodium chloride is not effective in removing the lysed cellular material. With an abundance of material deposited on the cloth, the SLS is responsible for the cleansing occurring in the solution. The same trend can be seen in the 2.0% NaC1/SLS solution (Figure 2). The cleansing remains steady throughout the solutions, and does not drop off as much as in the 0.9% NaCI. With more Na + and CI- in solution, more SLS is required to create micelles because the sodium chloride restricts micelle formation. The sodium chloride can sus- pend the cells and distribute them into the solution, and the SLS lyses these cells and removes the denatured cellular material. Although the micelies can lyse the cells at low concentrations of SLS, there are not enough micelies to adequately cleanse the denatured
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)