LG106W AND INCLUSION COMPLEXES 237 (7) (8) (9) (10) (ll) (12) (13) (14) 05) (16) (17) (18) H. H. Lee, Development of whitening agents by synthesis of polyhydroxy aromatic compounds, 3rd ASCS Scientific Conference, Taipei, 1997. T. Higuchi and J. L. Lach. Investigation of some complexes formed in solution by caffeine. IV: Interaction between caffeine and sulfathiazole, sulfadiazine, p-aminobenzoic acid, benzocaine, pheno- barbital and barbital, J. Am. Pharm. Assoc., Sci, Edn., 43, 349-354 (1954). T. Higuchi and K. A. Connors. Phase-solubility techniques, Adv. Anal. Chem. Instr., 4, 117-212 (1965). R. Lotan and D. Lotan, Stimulation of melanogenesis in a human melanoma cell line by retinoids, Cancer Res., 40, 3345-3350 (1980). T. Mossman, Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxity assays, J. Immunol. Meth., 65, 55-63 (1983). K. Uekama, Enhanced dissolution and oral bioavailability of ct-tocopheryl esters by dimethyl-[3- cyclodextri,n complexation,J. Inc. Phenom., 6, 167-174 (1988). J. Szejdi, E. Bolla-Pusztai, and M. Kajatar, The [3-cyclodextrin inclusion complex of menadione (Vitamin ,K3), Pharmazie, 37, 725-728 (1982). J. Szejtli, E. Bolla-Pusztai, M. Tardy-Lengyel, P. Szab6, and T. Ferenczy, Preparation, properties and biological activity of [3-cyclodextrin inclusion complex of menadione, Pharmazie, 38, 189-193 (1983). Takeda Chemical Industries Limited, Cyclodextrin inclusion compound of 2-n-octyl-4-octyl- isothiazoline-3-one useful as a fungicide having long-lasting effect, Japanese Patent 1505 77 (1983). Sunstar Inc., Nonirritating cosmetics, Japanese Patent 10323 (1984). Sun Star Dentifrice, Dentifrice composition without lowering the potency, Japanese Patent 1101115 (1976). Sun Star Dentifrice and Daiichi Kogyo Seiya (S. Hashimoto, K. Inoue, and T. Watanabe), Toothpaste compositions containing sucrose fatty ester foaming agents and cyclodextrin to prevent decrease in foaming ability without inactivating enzymes present, Japanese Patent 2034941 (1977).
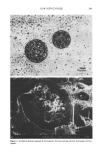
j. Cosmet. sci., 51,239-252 (July/August 2000) Preparation of a new soft capsule for cosmetics K. MIYAZAWA, I. YAJIMA, I. KANEDA, and T. YANAKI, Basic Research Center, Shiseido Co. Ltd., 1050 Nippa-cho, Kohoku-ku, Yokohama, Kanagawa, 223-8553Japan. Accepted for publication July 10, 2000. Presented in part at the Annual Meeting of The Pharmaceutical Society of Japan, Gifu, Gifu Pref., Japan, March 29, 2000. Synopsis A novel microcapsule for cosmetics was studied. The microcapsule was prepared by using an O/W/O emulsification technique and showed high thermostable properties (70øC). Agar was employed to gel the water phase of the emulsion. The size of the microcapsules was controlled by the process temperature and the stirring speed: the higher the temperature or the stirring speed, the smaller the diameter of the microcapsule produced. The strength of the agar gel phase was investigated, and the effect of the ratio of the internal oil to the gel (water) phase was observed. Young's modulus decreased with increase of the internal oil ratio. For cosmetic applications of the microcapsules, the all trans-retinol palmirate or ethyl linoleate used as a model ingredient was stabilized by microencapsulation. The remaining percentages of all trans-retinol palmirate and ethyl linoleate in the microcapsule after the four-week experiments at 50øC were 87% and 95%, respectively. On the other hand, those in oil solution were 72% and 91%, respectively. This micro- capsule shows suitable properties for not only cosmetic use, but also for applications in foods and other products. INTRODUCTION Microcapsules are used in food industry (1,2), medical treatment (3-6), printing (7), cosmetics (8-10), etc. The methods of preparation are roughly divided into three types: (a) chemical (polymerization) (11-13), (b) physiochemical (coacervation) (14-18), and (c) physical (spray-drying and employing double-cylinder equipment) (19,20). The main purposes of microencapsulation are the protection of core substances from the surround- ing environment and the control of internal agent release (21). The biological compat- ibility and the excellent texture of the capsule are also the most essential factors for its application to cosmetics. The current microcapsules for cosmetic use are mainly prepared by a coacervation method using gelatin (22-24) or various other polymers (25-27). However, there is a disadvantage to employing the coacervation method. Because the membrane of microcapsules prepared by the coacervation method is vulnerable, glutar- aldehyde treatment is essential to cross-link gelatin for enhancing the strength of the capsule membrane. As a result, a complicated washing process has to be introduced to 239
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)