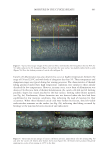
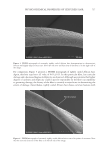
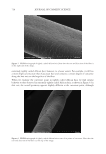
696 JOURNAL OF COSMETIC SCIENCE REFERENCES (1) J. M. Marsh, A. Schwan-Jonczyk, M. G. Davis, and S. W. Slofel, Consumer Perception versus Single and Bulk Fiber Technical Measurement, Weimar, Germany, 2009 International Hair Science Symposium. (2) C. R. Robbins. Chemical and Physical Behavior of Human Hair. 3rd Ed. Springer-Verlag, New York, 1994. (3) L. Kreplak, C. Merigoux, F. Briki, D. Flot, J. Doucet, Investigation of Human Hair Cuticle Structure by Microdiffraction: Direct Observation of Cell Membrane Complex Swelling, Biochim. Biophys. Acta., 1547(2), 268–274 (2001). (4) M. Gamez-Garcia, Cuticle De-cementation and Cuticle Buckling produced by Poisson Contraction on the Cuticular Envelope of Human Hair, J. Soc. Cosmet. Chem. 49, 213–222 (1998). (5) M. Gamez-Garcia, Patterns of Light Interference Produced by Damaged Cuticle Cells in Human Hair, J. Cosmet. Sci., 58(4), 269–282 (2007). (6) M. Gamez-Garcia, The Effects of Lipid Penetration and Removal from Subsurface Microcavities and Cracks at the Cumin Cuticle Sheath, J. Cosmet. Sci., 60(2) 85–95 (2009). (7) M. Feughelman, Mechanical Properties and Structure of Alpha-Keratin Fibers, Wool Human Hair and Related Fibers. University of New South Wales Press, Sydney, 1997.
697 Address all correspondence to Jennifer M. Marsh, marsh.jm@pg.com UV Oxidation: Mechanistic Insights Using a Model System JENNIFER M. MARSH, STEPHANIE L. DAVIS, RUI FANG, MONIQUE S. J. SIMMONDS, PHILIP GROVES AND VICTOR CHECHIK The Procter & Gamble Company, Mason Business Center, Mason, Ohio, USA (J.M.M., S.L.D.) Royal Botanic Gardens, Kew, Richmond, Surrey, United Kingdom (R.F., M.S.J.S.) Department of Chemistry, University of York, Heslington, York, United Kingdom (P.G., V.C.) Synopsis Damage to hair by UV radiation is relevant to most people, and for many, it is a major source of hair damage. The chemistry is complex, and studying all of the detailed reactions is extremely challenging. The objective of this work was to create a model system that allows for the study of one key mechanism involved in UV damage: the oxidation of tyrosine. A colloidal system to study the reactivity and chemistry of tyrosine was tracked by monitoring tyrosine decomposition and dityrosine formation by fluorescence spectroscopy. Experiments showed both the important role of oxygen in the decomposition of tyrosine and how the addition of redox metals such as iron can accelerate this decomposition. Finally, an antioxidant, butylated hydroxytoluene, was demonstrated to reduce this oxidation through the interception of reactive oxygen species. These findings suggested two possible strategies for reducing UV-induced radical damage to hair: removal of redox metals and addition of a radical scavenger to react with reactive oxygen species formed. The second strategy was confirmed by testing selected extracts from tea (Camellia sinensis), which is well known to have antioxidant properties. An oxygen radical antioxidant capacity assay was used as a screening tool to identify which tea extracts have the highest antioxidant efficacy, and these extracts were shown to reduce UV-induced protein damage in hair. INTRODUCTION Healthy hair is described as hair that has shine, can be readily styled, and has low levels of frizz in high humidity. In contrast, damaged hair shows signs of breakage, lack of alignment, split ends, and frizz (1). This difference is driven by multiple insults including oxidative colorants, UV exposure, heat, and physical damage from grooming habits such as washing and combing. Exposure to too much sun leading to lighter hair and damaged tips is a well-recognized issue for women, and breakage is often observed after prolonged exposure. Degradation of different parts of the hair structure by UV radiation has been well-documented in the literature, with changes to cuticle and cortex protein structures (2), cell membrane lipids (3), and melanin (4). J. Cosmet. Sci., 72, 697–710 (November/December 2021)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)